Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Co-Cultures of Caco-2 cells and E.coli
Optosensoric online monitoring of O2 uptake and extracellular acidification
Martin Brischwein and Daniel Grundl
Heinz-Nixdorf-Lehrstuhl für Medizinische Elektronik, TU München
Oligogalacturonic acids (OGAs), obtained by hydrolysis of pectin, are known to inhibit the adhesion of pathogenic bacteria on intestinal cells and thus to prevent infectious diseases. To test the effects of OGAs, an in-vitro model consisting of Caco-2 cells (human small intestinum) and the E.coli C25 strain was established. The co-cultures were set up in a novel, label-free cell assay platform working with a 24-well plate. The SDR SensorDish® Reader allows to monitor oxygen and pH kinetically in each well with chemical optical sensors to analyse the rates of cellular oxygen uptake and extracellular acidification.
Instrument and Sensors
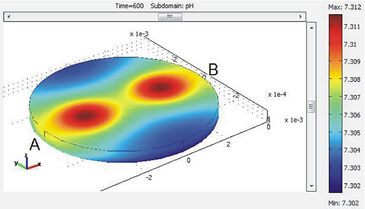
The instrument comprises a pipetting robot, climate box and sensor systems. The robot supplies media and active agents to the cells cultured in 24-well test plates (Fig. 1). The climate box provides constant temperature, humidity and aseptic conditions during long-term measurements. Chemical-optical sensors for dissolved oxygen and pH (PreSens GmbH) are excited / read out with a combined SDR SensorDish® Reader (PreSens GmbH) beneath the test plate for both parameters. The sensors are spotted onto the glass substrate forming the bottom of the plate. The readout frequency of each sensor is at 0.16 Hz. Using the cover lid of the test plate, small volumes of approx. 20 µl are adjusted in each well (chamber height approx. 0.5 mm) in order to achieve fast declines of oxygen and pH when the flow is stopped (approx. 10 min.). In the following flow cycle, the medium inside the microvolume is (partially) exchanged by the robot to recover the basic values. In time intervals of 2 hours, the medium inside the wells is exchanged completely. Rates of cellular oxygen consumption and extracellular acidification within the microvolumes are calculated from the slopes, i. e. ( Δ % O2/ Δt) and (Δ pH/Δt) during the time intervals without flow. Raw data evaluation (and system engineering) is further supported by process simulation with finite element (FEM-) numerical simulation, describing molecular diffusion, buffer capacity (Fig. 2) and liquid flow.
Metabolism of Caco-2 / E.coli Co-Cultures
The working hypothesis behind this work is (1) that E.coli C25 will cause detectable effects on Caco-2 metabolism upon adhesion and (2) that such effects can be inhibited by addition of OGAs. To test this hypotheses, Caco-2 cells were seeded on the test plate and cultured for 14 days to obtain a well differentiated layer of intestinal cells. Then the plate was inserted into the system platform and the measurement of pH and oxygen was started. After 2 hours, E.coli C25 cells were added to the storage vessel containing the medium, incubated for 1 hour and washed out again by the pipetting robot. Added bacteria were either pre-incubated with OGA (fraction V 12.1) or not. After washing out supernatant bacteria, ampicillin was added to the medium to prevent uncontrolled growth of remaining adherent bacteria. Data acquisition lasted 22 hours.
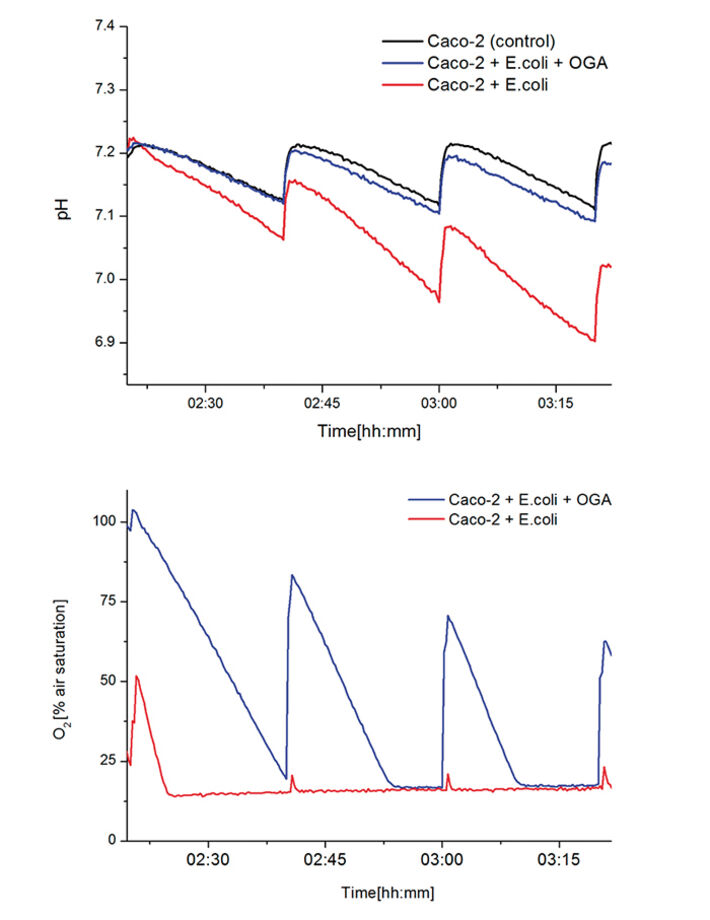
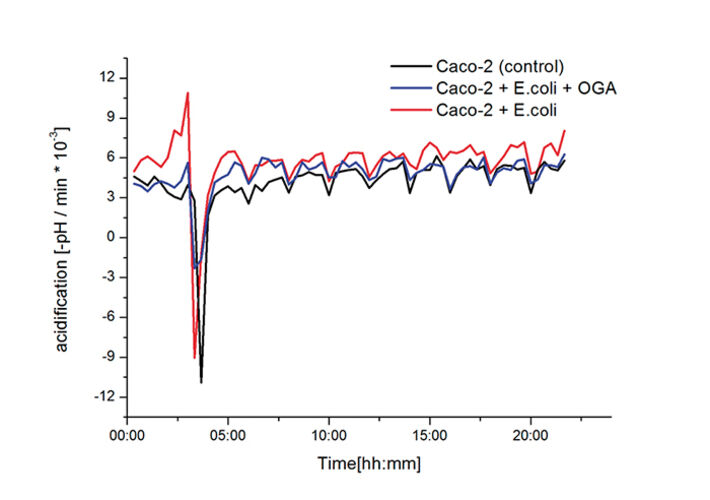
Fig. 3 shows the kinetics after switching from standard medium for the cell cultivation to 3 different media - cell culture medium for the Caco-2 cells (control), medium with E.coli and medium with E.coli with OGAs - but before washing out of supernatant bacteria. Comparing samples of E.coli with and without OGAs indicate that OGAs have - beyond an anticipated effect on adhesion - a clear growth inhibiting / cytotoxic effect on bacteria. Although at this stage of the assay, the same amount of E.coli is present in both media, the samples containing OGAs show much less activity than the samples with just E.coli. Comparing the pH kinetics, the samples with OGAs show only a slightly higher activity compared to the control without bacteria (indicated by the decreasing shifts at medium change). Recent results suggest that various fractions of OGAs obtained by pectin hydrolysis markedly differ in their adhesion-inhibiting and growth-inhibiting effects. In contrast to the working hypothesis however, it turned out that co-culturing of E.coli C25 on Caco-2 cells does not cause obvious effects on Caco-2 metabolism. Fig. 4 shows the entire kinetics of the assay. After washing out the supernatant bacteria after 4 hours, the acidification rates of the 3 differently treated samples do not differ largely.
The statistical evaluation including all wells (8 wells for each of the three groups with (1) Caco-2 cells alone, (2) Caco-2 + E.coli C25 and (3) Caco-2 + E.coli C25/OGA) confirms the absence of detectable effects on Caco-2 metabolism, which might be correlated with E.coli adhesion. Adhesion of E.coli C25 on Caco-2 cells itself was verified independently by counting colony forming units from adherent cells (data not shown). Obviously the pathogenic mechanism does not involve marked metabolic responses of the affected Caco-2 cells. The study now focuses on quantification of growth-inhibiting/toxic effects of the different OGA fractions on bacterial strains alone.
Conclusion
OGAs, obtained from pectin, are discussed as targeted food additive for their potential to treat diarrhea. The origin of this discussion was the realisation that OGAs are the effective component of carrot juice, which is - since 1908, when the pediatrist Dr. Moro discovered the effect - therapeutically applied to children suffering from diarrhea. The mechanism turned out to be an inhibitory effect of OGAs on the adhesion of pathogenic bacteria on intestinal cells. Targeted use of OGAs belongs to the area of "functional food", gaining increasing interest from food industry. This study, initiated in cooperation with the "Deutsches Institut für Lebensmitteltechnik" in Quakenbrück, Germany, is a contribution to create new scientific evidence for the existence (or non-existence) of benefits which may be expected from such additive food compounds.



