Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Feedback Controlled Carbon Feeding at Shake Flask Scale
Online measurement of DO with chemical optical sensors by PreSens

Manuela Senn and Martin Ischebeck
DSM Nutritional Products GmbH, Grenzach-Whylen, Germany
In this study a feedback controlled scale-down model at shake flask scale has been developed. Dissolved oxygen (DO) concentration in cultures of Saccharomyces cerevisiae was measured with chemical optical sensors (type PSt3) integrated in the shake flasks. The DO level triggered carbon feed on demand. This set-up allows automated fed-batch cultures in shake flasks and increases the quality of data obtained at this scale without compromising the quantity of results. Different strains in shake flask culture showed the same ranking as at bioreactor scale. In a second experiment a DO-stat and carbon limited process was tested in the feedback controlled system. Results showed that this process could be conducted successfully at shake flask scale with this set-up.
In bioprocess development for the production of pharmaceuticals, proteins, or nutritional additives scale-down models are used to save time and costs when testing for suitable cell lines, media composition, and process conditions. However, it is often difficult to determine critical parameters for scale up of processes at small scale. Shake flasks are commonly used in biotechnological research and production. They represent a low cost alternative to expensive larger scale bioreactor systems, and are a lot easier to handle. On the other hand there is no established system for online monitoring and control of important culture parameters at this scale. A reliable method for automated feeding and the elimination of manual sampling would be needed to improve bioprocess development at shake flask scale. In these experiments a feedback controlled system was tested that could be applied for shake flask cultures. A feed with carbon source was triggered by the DO content in the cultures, measured online with chemical optical sensor technology by PreSens. The oxygen sensors were integrated at the bottom of glass Erlenmeyer flasks and read out non-invasively through the transparent flask wall. The sensor signal was transferred to the OXY-10 mini, a 10-channel oxygen meter allowing simultaneous online measurements in up to 10 shake flasks. Using peristaltic pumps the glucose solution was added to the cultures automatically on demand. Tests with pulsed feed on demand in cultures of S. cerevisiae were carried out and results compared to values gained at bioreactor scale (20 L). Furthermore, the system was used to test a carbon limited process, used to enhance biomass and product yield, where DO was tried to be kept at around 20 %.
Materials & Methods
Experiments were carried out in 500 mL glass shake flasks with 100 mL working volume and a shaking rate of < 250 rpm at 2.5 cm throw, or < 200 rpm at 5.0 cm throw. Each flask had an oxygen sensor spot attached to its bottom, and was placed on top of the Coaster CFG. The Coaster is an accessory that allows transferring the sensor signal to the oxygen meter. The OXY-10 mini was connected via RS232 to a Lantronix S2E-Converter, which was connected to an AdamBus A/D-Converter. The AdamBus A/D-Converter controlled the peristaltic pumps for feeding. A PC with Lucullus PIMS was used to gather data from the Lantronix S2E-Converter and monitor the process. Each shake flask was connected to an individual peristaltic pump with silicone or pharmed tubing (12 - 16 tube diameter) allowing a flow of 0 - 50 g/h. The tubes were attached to the shake flask lids (see Fig. 1). The actual feed rate was calculated by means of a correlation of flow rate vs. pump speed. First, experiments with four different strains of S. cerevisiae were carried out in duplicate in 8 shake flasks. 500 g/L glucose solution were given to the cultures on demand as carbon source with pulses of 0.5 hours at a feed rate of 20 g/h. Feed rate and pulse time are experience values. Feed start was triggered when ΔDO/10 min was > 10 %. In a second experiment S. cerevisiae was tried to be kept at static DO in a carbon limited process. 150 g/L glucose solution was used and feed start triggered again when ΔDO/10 min was > 10 %. A simple 2-step controller was used to keep DO at a setpoint of 20 % with 0.5 % increment.
Results
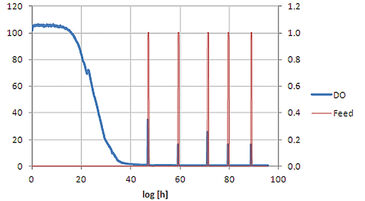
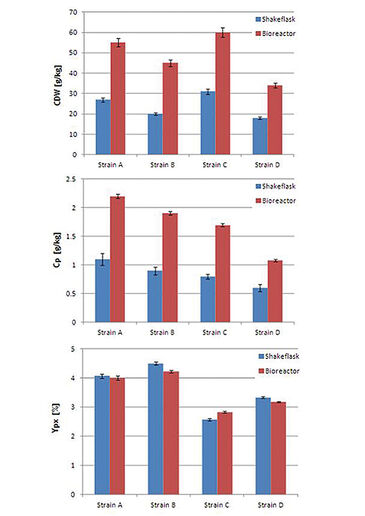
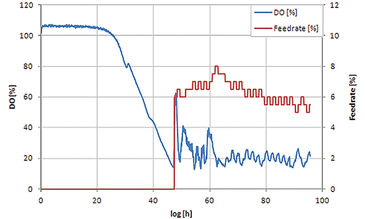
The feed on demand experiment was run for 96 hours (see Fig. 2). In the first phase the initial glucose is consumed and due to the diauxic growth of S. cerevisiae partly converted to ethanol. The growth of the cells leads to a decrease of DO level and at around 22 h (small peak in DO signal) glucose is depleted and growth continues on ethanol. Due to the limited oxygen transfer capacity of shake flasks the DO level reaches zero at around 40 h. When ethanol is also depleted, no other carbon source is present in the medium resulting in reduced oxygen uptake by the cells and therefore an increase in DO level. This increase is used as trigger for glucose addition. As soon as the added glucose and the produced ethanol are consumed again, the next additon is started. Comparing dry weight and production yield of the four S. cerevisiae strains in the shake flask set-up with results gained for the same strains at bioreactor scale (20 L) it turned out that the shake flask cultures reached lower output in cell dry weight and product concentration (Fig. 3), which is caused by the higher oxygen transfer capacity of bioreactors. Nevertheless, all strains showed the same ranking at both cultivation scales, which enables picking strains with special characteristics for later scale-up processes. Comparison of relative production parameters revealed good alignement of the shake flask culture values with those taken at bioreactor scale (Fig. 3). In a second experiment the automated shake flask system was tested in carbon limited cultivation with static DO. Fig. 4 shows that the automated feeding system could be used to perform DO-Stat cultivation. When DO levels fall below setpoint feedrate is decreased and vice versa. The chosen parameters (0.5 % feedrate increment, 1 h step time) already resulted in a very good DO control. Further fine tuning with defined process conditions can lead to even lower oscillation in the DO signal.
Conclusion
The developed feed-on-demand system for shake flask cultivation showed good results and compared with values from bioreactor scale it can be concluded that different strains can be compared and picked at this scale. Moreover tests in this set-up were extremely time saving. Where only 2 or 3 experiments can be run at bioreactor scale, this systems allows conducting 8 - 10 tests a week. Even in a run with a carbon limited process the system performed with unexpectedly good results. Being able to measure DO online and in several shake flasks simultaneously with the PreSens OXY-10 mini is an important advantage, when conduction experiments in scale-down processes. Further experiments with the SFR Shake Flask Reader that allows for oxygen and pH measurement in up to 9 shake flasks might enhance this automated and partly controllable system at shake flask scale. Further development of the set-up is already planned.