Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Investigation on ATP Synthesis via Oxidative Phosphorylation in C. glutamicum
Measurement of oxygen consumption by an ATP synthase deletion mutant and the wild type strain with the SFR Shake Flask Reader

A. Koch-Koerfges1, A. Kabus1, I. Ochrombel2, K. Marin2, and M. Bott1
1Institute for Bio- and Geosciences (IBG-1), Forschungszentrum Jülich, Germany
2Institue for Biochemistry, University of Cologne, Germany
A F1F0-ATP synthase deletion mutant of C. glutamicum was characterized and investigated in respect to growth, by-product formation and bioenergetics. The mutant did not grow with acetate as sole carbon source but reached 47% of the growth rate and 65 % of the biomass of the wild type when cultivated in shake flasks with glucose as sole carbon and energy source. Dissolved oxygen (DO) in the culture was monitored online, and non-invasively with the SFR Shake Flask Reader, which allowed to compare oxygen consumption of wild type and mutant strain with analysis results of other parameters. The SFR measurement displayed the reason for a biphasic glucose uptake rate of the mutant: when oxygen became limiting, the glucose consumption rate of the mutant was reduced below that of the wild type. The results show that F1F0-ATP synthase and oxidative phosphorylation are in general not essential for growth of C. glutamicum on substrates allowing substrate level phosphorylation.
C. glutamicum is the most important microorganism for large scale biotechnological production of L-glutamate and L-lysine, and also serves as a model organism for several pathogens, e. g. Mycobactericum tuberculosis, closely related to it. Therefore the metabolism of this bacterium is intensely studied. Most aerobic, respiring bacteria gain ATP both by substrate level phosphorylation (SLP) and by oxidative phosphorylation, or more generally electron transport phosphorylation (ETP). In this study the role of ATP synthesis in C. glutamicum by ETP was investigated closer. In ETP the microorganism synthesizes ATP with the membrane bound F1F0-ATP synthase using the proton motive force as driving force. Though it is generally assumed that C. glutamicum synthesizes ATP mostly by ETP hardly any tests on the F1F0-ATP synthase have been done yet. So in this study a defined ΔF1F0 mutant of C. glutamicum strain ATCC 13032 lacking all genes encoding the F1F0-ATP synthase was analyzed. The strains were cultivated in shake flasks with integrated chemical optical sensors, and non-invasive, online monitoring of DO was conducted with the SFR Shake Flask Reader by PreSens. This way oxygen consumption in cultures of the mutant and the wild type could be measured over the whole cultivation time and later compared with analysis results for other parameters.
Materials & Methods
C. glutamicum strains and plasmids used in this work as well as preculture conditions are listed and described in [1]. Main cultivations were done in baffled 500 mL glass shake flasks containing a septum for sterile sampling with 50 mL glucose minimal medium at 30 °C and 130 rpm. Oxygen consumption was determined using the SFR Shake Flask Reader (PreSens) based on non-invasive measurement with autoclavable chemical optical sensors. These sensors are integrated in the shake flasks (Fig. 1) and read out through the transparent flask wall with the SFR, which is integrated in the shaker. DO in % air saturation was measured at an interval of 10 min throughout cultivation time. Methods for determining growth rate, biomass concentration, and glucose as well as organic acids are described in detail in [1]. Specific glucose uptake rates (sGUR) were calculated using the equation:
sGUR (ti,ti+1) = ((cglc,i - cglc,i+1)/(ti+1 - ti)) x
(2 / (XCDW,i+1 + XCDW,i)) [gglucose/gDW h]
cglc represents the glucose concentration (g L-1), XCDW the cell dry weight (CDW) per volume (g L-1) and (ti+1 - ti) the time interval (h) investigated.
Results
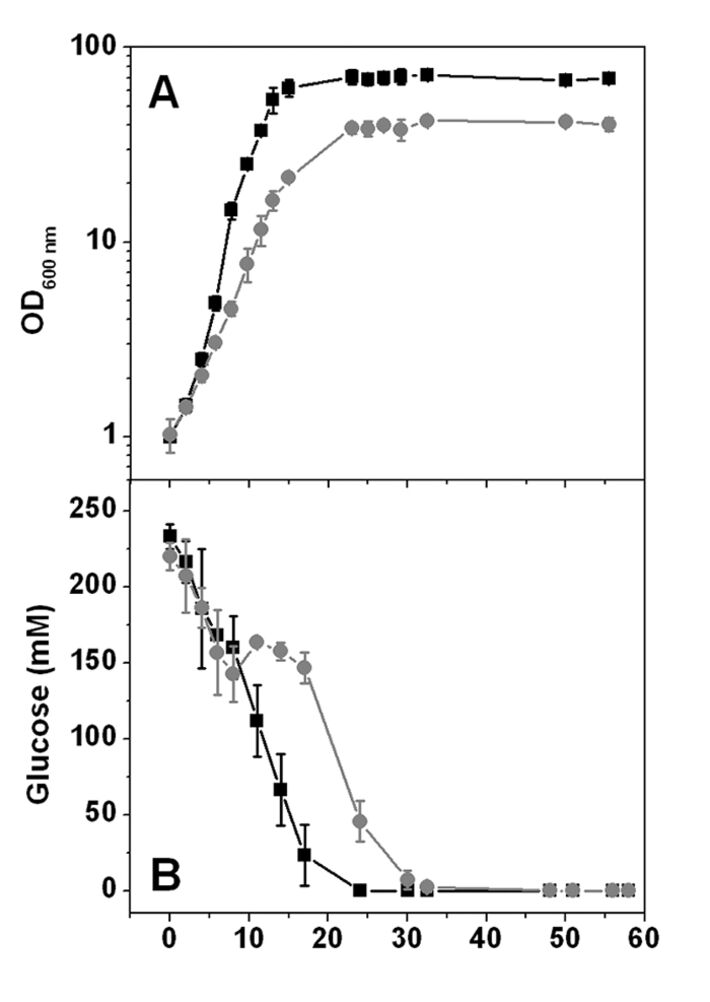
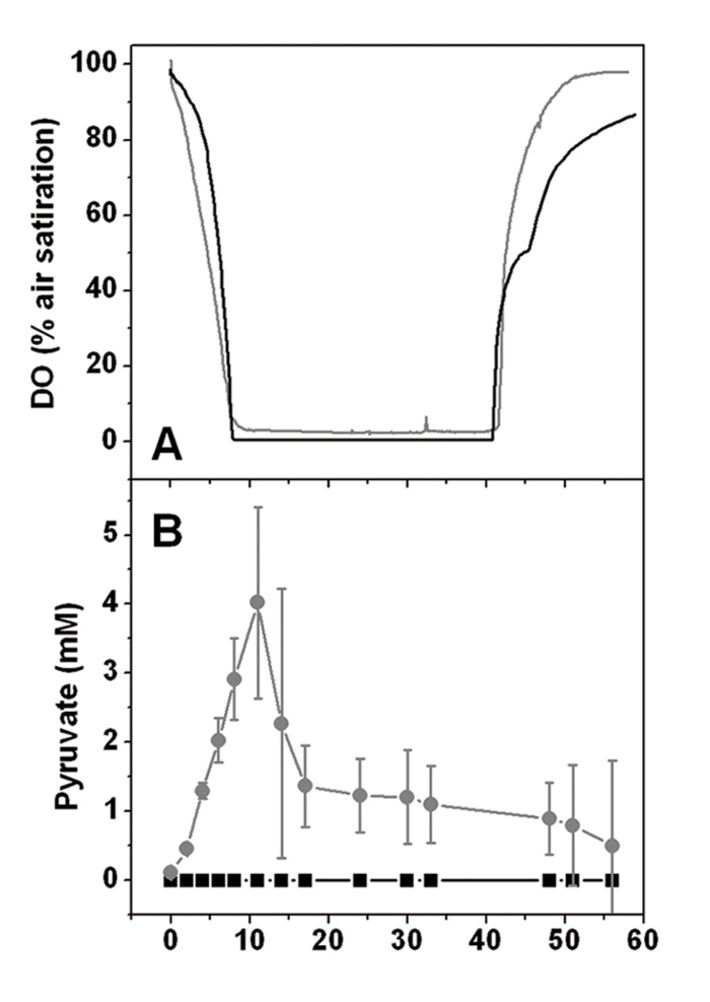
Initial tests on agar plates containing CGXII minimal medium with either 222 mM glucose or 100 mM acetate as sole carbon source were conducted. It turned out that the mutant was able to grow with glucose, much slower than the wild type though, but it could not grow with acetate as sole carbon source. In contrast to glucose, acetate catabolism in an ATP synthase mutant does not allow net ATP synthesis via SLP. In shake flask culture using CGXII minimal medium with 4 % (w/v) glucose as sole carbon and energy source the ΔF1F0 mutant was able to grow with 47 % of the growth rate of the wild type (0.19 ± 0.02 h-1 vs. 0.40 ± 0.01 h-1) and reached 65 % of the biomass of the wild type (10.3 ± 0.5 g (CDW) L-1 vs. 15.8 ± 3 g (CDW) L-1), as displayed in Fig. 2 A. These results reveal that the F1F0-ATP synthase is not essential for the growth of C. glutamicum on substrates allowing net ATP synthesis via SLP. The wild type culture continuously took up glucose until it was completely consumed after 24 h (Fig. 2 B). The kinetics of the ΔF1F0 mutant on the other hand showed two distinct phases with a change after 5 - 11 h, and glucose consumption was finished after 30 h. Within the first hours of cultivation the calculated sGUR of the mutant reached values of ≥ 328 ± 72 nmol min-1 (mg CDW)-1 which are threefold higher than for the wild type with sGUR ≥ 89 ± 1 nmol min-1 (mg CDW)-1. In the second phase, the mutant´s sGUR values were in the range of 21 ± 16 nmol min-1 (mg CDW)-1, so much lower than in the wild type. In order to find an explanation for the biphasic glucose uptake of the mutant, oxygen consumption was measured by optical sensing of dissolved oxygen (DO) with the SFR. Initially the ΔF1F0 mutant consumed oxygen faster than the wild type (Fig. 3 A) while showing a lower growth rate, which suggests a much higher respiration rate of the mutant. DO concentration in the mutant culture reached 0 after 7 - 8 h. Oxygen depletion correlated with the decrease in glucose uptake rate. So the availability of the terminal electron acceptor oxygen caused the two distinct phases in the mutants' glucose catabolism. The increased glucose consumption was accompanied by the excretion of pyruvate in the medium right from the start of the cultivation (Fig. 3 B), while no other organic acids could be detected. After 8 h, when oxygen became limiting and the glucose uptake rate was reduced, pyruvate was consumed again. In contrast to the ΔF1F0 mutant the wild type did not secrete pyruvate.
Conclusion
Investigations on a mutant of C. glutamicum lacking ATP synthesis via oxidative phosphorylation showed that it was able to grow on substrates like glucose allowing SLP. This proves that F1F0-ATP synthase is in general not essential for this organism. The glucose catabolism of the mutant showed two distinctive phases depending on the availability of the final electron acceptor oxygen. Conducting non-invasive, online monitoring of DO with the SFR made it easy to compare carbon source consumption with oxygen uptake of the mutant and wild type. The SFR proved to be an easy and time saving tool, allowing to gain important insight in the metabolism of this mutant strain.
Application note adapted from
[1] A. Koch-Koerfges et al., 2012, Physiology and global gene expression of a Corynebacterium glutamicum ΔF1F0-ATP synthase mutant devoid of oxidative phosphorylation, Biochimica et Biophysica Acta 1817, pp. 370 - 380


