Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Oxygen Dynamics in the Capillary Fringe
Mapping oxygen consumption of Pseudomonas fluorescens with VisiSens
Norman Hack & Harald Horn
Chair of Water Chemistry and Water Technology, Engler-Bunte-Institut, Karlsruhe Institute of Technology, Germany
The capillary fringe is a highly active zone for chemical and biological transformation at the transition of the vadose zone and groundwater. To study aerobic biodegradation in the capillary fringe a Hele-Shaw-Cell was used in these experiments. Oxygen sensor foils were integrated in the cell for non-invasive mapping of metabolic activity with VisiSens. Tests were conducted with and without P. fluorescens, and furthermore with simulated groundwater flow. Without the microbial strain no change in oxygen concentration could be measured, while in presence of P. fluorescens aerobic biodegradation of the model compound could be clearly detected in the oxygen maps recorded over time. In the experiments with simulated groundwater flow the biological activity and oxygen consumption was even higher.
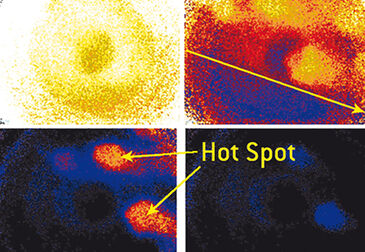
Due to the broad abundance of synthetic organic micro-pollutants, their behavior, dispersion and biodegradability is of great importance for the understanding of biodegradation processes in the environment. In this subproject of the DyCap (dynamic capillary fringe) research group, the main objective is to study mass transfer and biological transformation. Experiments with organic compounds (e. g. salicylic acid, X-ray contrast media) in batch (diffusion), in column (vertical flow) and in a flow-through cell (horizontal and vertical flow: 2-dimension flow-through microcosm) systems / set-ups are conducted. The concentration of dissolved oxygen plays an important role in biodegradation. Aerobic microorganisms in the capillary fringe (CF) use the dissolved oxygen to oxidize synthetic organic micro pollutants. Oxygen is an electron acceptor for (bio) chemical redox reactions and therefore an indicator for the biological activity. In former experiments dissolved oxygen was non-invasively detected by means of optrode techniques (Fibox 3, PreSens Precision Sensing GmbH). The oxygen stripes have the advantage of fast one-dimensional determination of oxygen. For a 2-dimensional visualization and development of oxygen profiles, VisiSens sensor foils (PreSens Precision Sensing GmbH) offer significant advantages. The time-lapse development of oxygen-rich and oxygen-poor areas can be visualized. Therefore, interfaces and hot-spots (captured zones with oxygen) are clearly determinable and examinable. With this data, e. g. biodegradation kinetics for freely definable zones or at certain points are assessable.
Materials & Methods
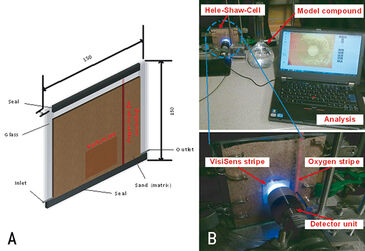
To simulate the natural soil matrix, a Hele-Shaw-Cell (2D; 150 x 150 x 10 mm3) was filled with quartz sand of defined grain size (dp = 200 - 600 µm) (Fig. 1). Hele-Shaw-Cell and tubes were sterilized with T = 121 °C for 1 hour to avoid other microbial strains. The change of air saturation over time was monitored in the Hele-Shaw-Cell under three different experimental conditions:
1. Inoculation up to 15 hours of the model compound without addition of P. fluorescens driven by capillary force (bottom to top).
2. Inoculation up to 30 hours of the model compound with added P. fluorescens driven by capillary force (bottom to top).
3. To simulate ground water flow behavior, the model compound was inoculated up to 24 hours through the inlet tube by means of a peristaltic pump (flow rate: 60 µL/min). P. fluorescens were inoculated step by step, in alternation with sand into the Hele-Shaw-Cell, to ensure equal distribution in the matrix.
To detect dissolved oxygen, a sensor stripe (for control; 10 x 20 mm2 read with the Fibox 3) and the VisiSens sensor foil (SF-RPSU4, 40 x 40 mm2) were used which allowed fast, in situ and non-invasive measurement. The oxygen ratio and concentration were measured every 10 minutes up to 30 hours. Dynamics in air saturation were derived from Fibox readings as well as from VisiSens time series measurement of dissolved oxygen.
O2 Dynamics in the Hele-Shaw-Cell
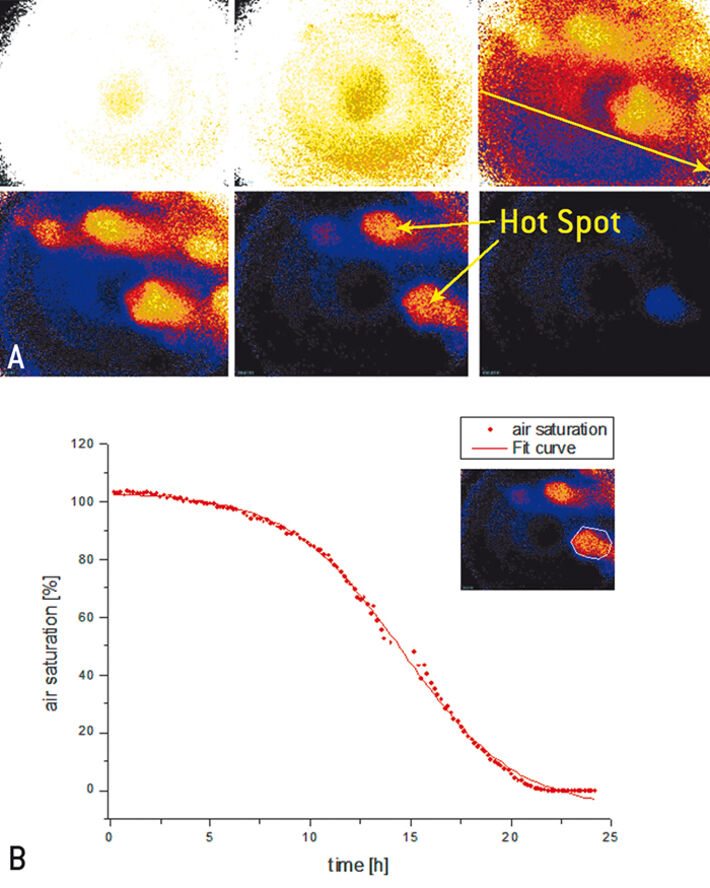
Without P. fluorescens and without simulated groundwater flow no change for the air / oxygen saturation was measured within 15 hours, as has been expected. The yellow colored area in Fig. 2 A corresponds to an air saturation of 100 % (ROI: 0.8205). The darker area in the middle of each picture seems to be an artifact due to reflection by camera LED lights. In presence of P. fluorescens and without simulated groundwater flow a change in oxygen saturation within 30 hours could be recorded (Fig. 2 B). The aerobic biodegradation of the model compound started after 10 hours. The P. fluorescens consume oxygen especially in the lower part (0 - 2 cm) and an anoxic zone (black colored, ROI: 1.29) was formed. Due to the experimental set-up, the concentration of P. fluorescens near the bottom of the Hele-Shaw-Cell is higher compared to upper regions (> 4 cm), since the transport is coupled to the capillary force. Furthermore, there is the possibility of adsorption on sand or clogging. Hence, the oxygen consumption rate is not evenly distributed across the Hele-Shaw-Cell (e. g. after 19 and 21 hours). Changes of oxygen saturation within 24 hours in presence of P. fluorescens and simulated groundwater flow are shown in Fig. 3 A. In comparison to experiment results without inlet and outlet flow and capillary action as only driving force the biological activity and oxygen consumption rates were much higher. The anoxic zone (ROI: 1.3065) reaches a thickness of 8 cm of the capillary fringe. It´s due to an even distribution of P. fluorescens concentration. The anoxic zone was formed from left to right with a decreasing gradient (indicated by the yellow arrow after 20 hours). This goes along with the continuously refreshing model compound concentration near the inlet port. After 21 hours different oxygen rich hot spots were formed. This is due to possible different bulk density of the matrix. Over the whole time period P. fluorescens consumed oxygen in these areas and the whole examined area became anoxic. Degradation kinetics of oxygen consumption was determined (via z-profile) and a fit curve was calculated (Fig. 3 B). The fitted curve follows the expected first order kinetic.

Conclusion
In three experiments oxygen uptake was determined using oxygen sensitive foils and the VisiSens imaging system. After adding a microbial strain (Pseudomonas fluorescens) and a model compound (substrate) the oxygen concentration decreased in the Hele-Shaw-Cell. The VisiSens foil showed the expected oxygen profile over time. VisiSens offers a lot of advantages especially the online measurement and the study of the locally resolved oxygen distribution. It makes the "oxygen profile vs. height of the capillary fringe" visible. The oxygen rich and oxygen poor areas, hot spots and also the interfaces are clearly distinguishable. It is easy to handle and a good alternative to single-point measurement such as Fibox stripes for fast in situ measurements.