Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Direct Measurements of pCO2 in the Digestive Tract of Fish
Use of fiber optic pCO2 microsensors to reliably measure in vivo pCO2 in various sections of the gastrointestinal system in several fish species
Chris M. Wood1,2, Junho Eom1,2, Ellen H. Jung1,2, and Fernando Martinez Ferreras3
1Department of Zoology, University of British Columbia, Vancouver, B. C., Canada
2Bamfield Marine Sciences Centre, Bamfield, B. C., Canada
3PreSens Precision Sensing GmbH, Regensburg, Germany
Our objective was to measure the pCO2 levels in the fluids of the stomach and various sections of the intestine in both fasted and fed fish, weighing typically 50 - 300 g. It was essential that these measurements be made without rupturing the tract, so as to prevent equilibration with the ambient media. Conventional pCO2 electrodes are simply too large to insert into the tract without severely damaging it, and in any event are not designed to function in a thick viscous slurry such as chyme (partially digested food mixed with gastrointestinal secretions). Needle-mounted PreSens fiber optic pCO2 microsensors proved to be ideal for this purpose, as the needle could be inserted through the wall of the tract directly into the desired location, allowing sequential monitoring of pCO2 in various sections of the gut. These measurements demonstrated that the pCO2 in the digestive tract is surprisingly high even in fasted fish, far above blood level, and is markedly elevated after feeding in teleosts. The digestive tract is extremely well vascularized with fine blood vessels, and receives a substantial portion of the cardiac output, so these observations raise interesting questions as to how the systemic blood is protected from equilibrating with these high pCO2 levels, which would otherwise cause severe respiratory acidosis and disturb blood O2 transport.
Because the solubility of O2 in water is very low relative to the solubility of CO2, the gills of fish are essentially hyperventilated with respect to CO2 excretion [1]. As a result, CO2 is washed out of the blood to very low levels, so blood pCO2 is typically just a few torr (i.e. less than 0.5 kPa or about 0.5 % CO2). This low pCO2 is critical in setting the correct pH in the blood and body fluids. However, there is now increasing concern about how fish will deal with the elevated environmental pCO2 levels predicted to occur with climate change by the end of the century (e.g. + 0.75 torr, + 0.1 kPa or about + 0.1 %; [5]), and about the much higher pCO2 levels that are already experienced by fish in intensive aquaculture (e.g. + 7.5 torr, + kPa, or about + 1 %; [2]). However in mammals, there exists evidence that the pCO2 in the digestive tract may be much higher than that in the blood. For example, pCO2 levels measured in human flatus were about 6-fold greater than normal blood pCO2 levels [8]. If the same is true in fish, then physiological mechanisms may already be in place to prevent this internal pCO2 threat from equilibrating with the blood and disrupting acid-base regulation and O2 transport. The PreSens fiber optic pCO2 microsensors provided a unique methodology to address this question.
Materials & Methods
Experimental Animals
Fish were obtained from either aquaculture (freshwater rainbow trout Oncorhynchus mykiss and goldfish Carassius auratus) or captured from the wild (seawater English sole Parophrys vetulus and hagfish Eptatretus stoutii), the latter under collection permits from the Department of Fisheries and Oceans Canada. The trout, goldfish, and sole are teleost fish, while the hagfish is an agnathan, an extant representative of the earliest craniates. The trout was chosen as a typical carnivore that has an acidic stomach, while the goldfish is a herbivore that completely lacks a stomach. The marine sole is also a carnivore with an acidic stomach, but is additionally known to secrete HCO3- in its intestine as part of its osmoregulation strategy in the marine environment [4]. The hagfish is a carnivore, and like the goldfish, lacks a stomach.
In the laboratory, the fish were kept at 12 °C and were either fasted such that the digestive tract was empty, or fed to satiation with commercial pellets (trout, goldfish), frozen herring (sole) and squid (hagfish). The fish were anaesthetized with neutralized MS-222 (Syndel Laboratories, Parksville, B. C., Canada), and in the case of trout and goldfish were implanted with a dorsal aorta catheter for arterial blood sampling. Each experimental animal was placed on an operating table and continuously irrigated (i.e. artificially ventilated) via the gills (trout, goldfish, sole) or nostril (hagfish) with temperature-controlled water (12 °C) containing MS-222 at a concentration appropriate for each species so as to maintain Stage 5 anaesthesia - "loss of reflex activity, total loss of reactivity, shallow opercular movements" [6]. The peritoneal cavity was surgically opened to expose the gastro-intestinal tract, which was kept moist throughout with physiological saline. Typical experimental periods were 3 - 5 h, and the fish continued to ventilate throughout.
pCO2 Measurements
Prior to each experiment, the needle-mounted fiber optic pCO2 microsensor was calibrated at the experimental temperature (12 °C) using 0.9 % or 3.5 % (hagfish) NaCl solutions equilibrated with a range of CO2-in-air mixes (typically 0.04, 0.1, 0.5, 1, 3 and 5 % and occasionally 10 %) created by a 301aF precision gas-mixing pump (Wösthoff Messtechnik GmbH, Bochum, Germany). The calibration procedure typically took about 1.5 h. Additional calibration checks were performed during and after experiments.
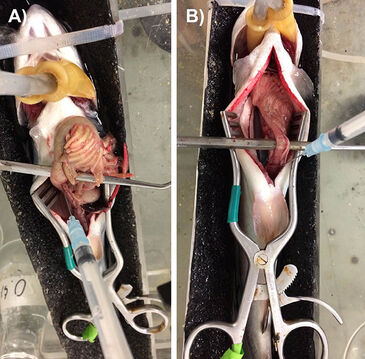
Each PreSens microsensor was mounted inside a 23Gx11/4" hypodermic needle connected to a 1 mL plastic syringe. The syringe was mounted on a M33301R three-axis micromanipulator (World Precision Instruments, Sarasota, FL, USA) so that the probe could be appropriately positioned. pCO2 measurements were made by inserting the needle-mounted microsensor into the desired section of the digestive tract, and then advancing the syringe plunger so that approximately 1 mm of the sensor tip was exposed. Fig. 1 illustrates the making of measurements in the mid-intestine of rainbow trout which had been (A) fed 24 h previously (note that the tract is engorged with chyme) or (B) fasted for > 1 week (note that the tract is empty). Additional pCO2 measurements were taken in blood samples (0.2 mL) freshly drawn from the arterial catheter (trout, goldfish) or taken by puncture of the caudal venous sinus in hagfish. Recordings were made at 2-min intervals, and final values were typically attained after 10 - 20 minutes when the probe trace reached a stable value.
Results and Discussion
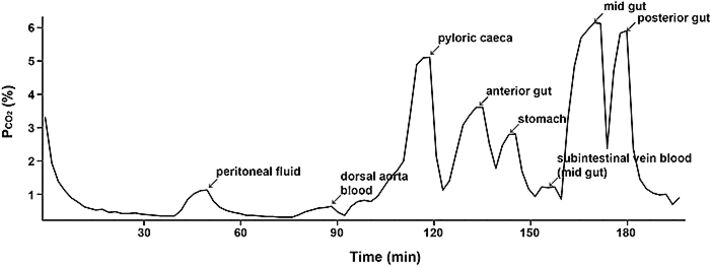
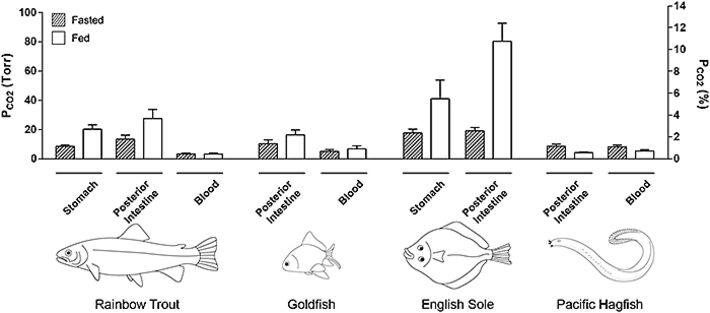
Figure 2 illustrates a representative experiment on a fed rainbow trout. It can be seen that pCO2 values varied greatly among recording sites, but that all values were markedly higher than in the blood. Data have been summarized for the four species for the stomach and posterior intestine (Fig. 3). Indeed even in fasted trout, mean pCO2 ranged from about 8.5 torr in the stomach to 13.4 torr in the posterior intestine in contrast to a normal blood pCO2 of only 3.3 torr. With feeding, the pCO2 values generally doubled or tripled throughout the tract, reaching means of 20.3 torr in the stomach and 27.4 torr in the posterior intestine, without any change in blood pCO2 (Fig. 3). In freshwater trout, the highest values occurred in the anterior intestine, surpassing 40 torr in some animals (data not shown), though not in all fish (e.g. Fig. 2). The agastric goldfish exhibited pCO2 values that were fairly uniform in the different sections of its very long intestine, but again these were two- to three-fold higher than blood levels, and tended to increase with feeding (Fig. 3)
With respect to seawater fish, measurements on the English sole are as yet preliminary (only two fed and two fasted animals, and no blood data so far), but they suggest that pCO2 levels in this marine teleost are markedly higher than in freshwater teleosts, reaching the highest values of about 80 torr in the posterior intestine of fed animals (Fig. 3). On the other hand, the "primitive" Pacific hagfish, which is agastric but with a very short intestine, exhibited completely different patterns from the other species. pCO2 values were uniform in all parts of the intestine, and these almost exactly matched blood pCO2. Curiously, rather than exhibiting increased digestive tract pCO2 after feeding, exactly the opposite occurred, with intestinal pCO2 falling in parallel to blood pCO2 (Fig. 3).
At present we are exploring the cause of these very high pCO2 levels in the digestive tract, and why they increase after feeding in the teleosts. Our findings to date suggest multiple pathways for CO2 generation in the teleosts, including the actions of gastric acid and intestinal enzymes on food components, the metabolism of the gut microbiome, the secretion of HCO3- by the pancreas and intestinal wall, and its subsequent reaction with acidic components in the chyme. In the marine sole, intestinal HCO3- secretion occurs at a very high rate, especially after feeding, reflecting the importance of this process in its osmoregulatory strategy [4], one shared with most other marine teleosts [3]. Hagfish on the other hand do not appear to secrete HCO3- into the intestine but do exhibit a vigorous increase in O2 consumption after feeding [9]. Perhaps the associated increase in ventilation explains the lower pCO2 levels after feeding.
Regardless, these findings raise exciting questions, in light of the extreme vascularity and high post-prandial blood flow ot the gastrointestinal tract [7]. Does the high pCO2 in the chyme equilibrate with the blood? Does it play a role in O2 delivery to the enterocytes by the Bohr and Root effects [1]? And if so, how is extreme respiratory acidosis in the systemic blood stream avoided? Preliminary measurements, such as the low pCO2 value recorded in the subintestinal vein of the trout shown in Fig. 2, suggest that equilibration may not occur, but if confirmed, this raises the further question as to how is it prevented? The PreSens fiber optic pCO2 (& pO2) microsensors will play important roles in addressing these questions.
References
[1] Dejours P. (1975), Principles of Comparative Respiratory Physiology. North-Holland Publishing Company, Amsterdam
[2] Good, C., Davidson, J., Welsh, C., Snekyik, K., and Summerfelt, S. (2010), Aquacultural Engineering 42, 51 - 56
[3] Grosell, M. (2011), Fish Physiology, Vol 30. (eds. Grosell, M. G., Farrell, A. P., and Brauner, C. J.) 135 - 164
[4] Grosell, M., De Boeck, G., Johannsson, O., and Wood, C. M. (1999), Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology 124, 259 - 270
[5] Heuer, R. M. and Grosell, M. (2014), American Journal of Physiology, Regulatory, Integrative, and Comparative Physiology 307, R1061 - R1084
[6] McFarland, W. N. (1959), Publ. Inst. Mar. Sci., 6, 23 - 55
[7] Seth, H., Axelsson, M., and Farrell, A. P. (2011), Fish Physiology, Vol. 30 (eds. Grosell, M. G., Farrell, A. P. and Brauner, C. J.) 351 - 393
[8] Suarez, F., Furne, J., Springfield, J., and Levitt, M. (1997), American Journal of Physiology - Gastronintestinal and Liver Physiology 272, G1028 - G1033
[9] Weinrauch, A. M., Clifford, A. M., and Goss, G. G. (2018), Journal of Comparative Physiology B 188, 101 - 112