Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Online Monitoring of Glucose, pH, and DO in Shake Flask Culture
Novel Glucose Sensor Used in Combination with the SFR Shake Flask Reader
I. Bauer1, G. T. John2, S. Spichiger3, and U. E. Spichiger-Keller3
1Zurich University of Applied Sciences, School of Life Sciences and Facility Management, Institute of Biotechnology, Biochemical Engineering and Cell Cultivation Technique, Waedenswil, Switzerland
2PreSens Precision Sensing GmbH, Regensburg, Germany
3C-CIT AG, Waedenswil, Switzerland
A new single-use glucose sensor in combination with chemical optical oxygen and pH sensors was used for online monitoring of CHO cell culture in shake flasks. During the whole process offline sampling was performed as well. The optimal point of harvest in the culture could be determined to be a lot earlier than it was stated by offline values so far. This study showed a difference in offline and online values caused by time difference and environmental conditions.
The FDA encourages manufacturers to use process analytical tools as effective and innovative approaches in development and quality assurance. The sensors described here allow enhanced monitoring of critical culture parameters during the process. Novel single-use glucose sensors (CITSens Bio, C-CIT, Waedenswil) make use of an enzymatic oxidation process and direct electron transfer from glucose to the electrode by a chemical wiring process. While other enzymatic glucose sensors show cross-sensitivity to oxygen this is not the case with the novel sensor type; to separate it from the cultivation medium, a dialysis membrane covers the sensing head. These sensors are integrated in the ventilation cap of shake flasks (Fig. 1). For the experiments described here the glucose sensors were used together with SFS Sensor Flasks by PreSens containing chemical optical oxygen and pH sensors. The optical sensors are read out non-invasively through the bottom of the flasks with the SFR Shake Flask Reader, which is installed inside the incubator. Both sensor systems allow wireless data transfer to a PC, so online data can be obtained without interferences, as opening the incubator door and taking out the shake flasks for sampling is not necessary.
Materials & Methods
Three 125 mL shake flasks with integrated pO2 and pH sensors were used in these experiments. Each was equipped with one of the glucose sensors by C-CIT integrated in the ventilation caps of the flasks. Cell culture medium (ChoMaster® HP-1, Cell Culture Technologies LLC) was inoculated with CHO cells and filled into the flasks. The starting viable cell density was 0.64 * 105 cells mL-1 at 97 % viability. Initial glucose concentration was 3.59 g L-1 measured with an enzymatic offline analyzer (BioProfile®, Nova Biomedical Corporation), and 4.05 g L-1 measured with HPLC. For practical reasons the glucose sensors were calibrated in a simple one point calibration according to the value of the enzymatic analyzer at 3.6 g L-1. Throughout the whole experiment samples were analyzed offline as well. For the first 4 days the cells were incubated at 37 °C to produce cell mass. After a media change to production medium on day 4 the temperature was reduced to 31 °C to induce protein production for the next 6 days.
Online vs. Offline Analysis Values
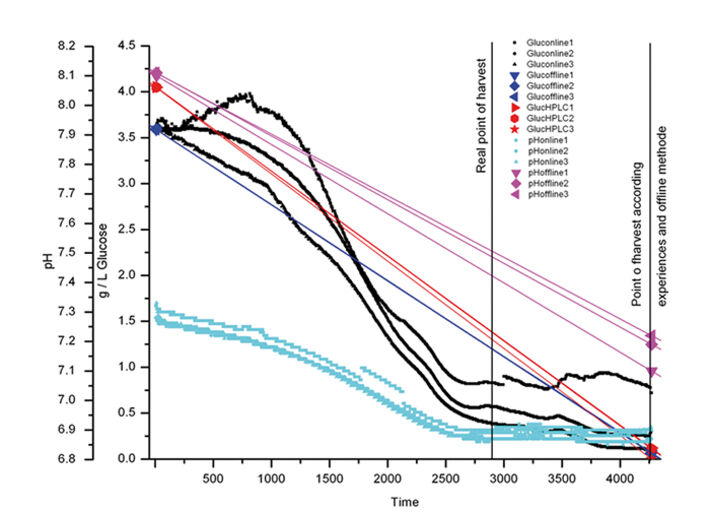
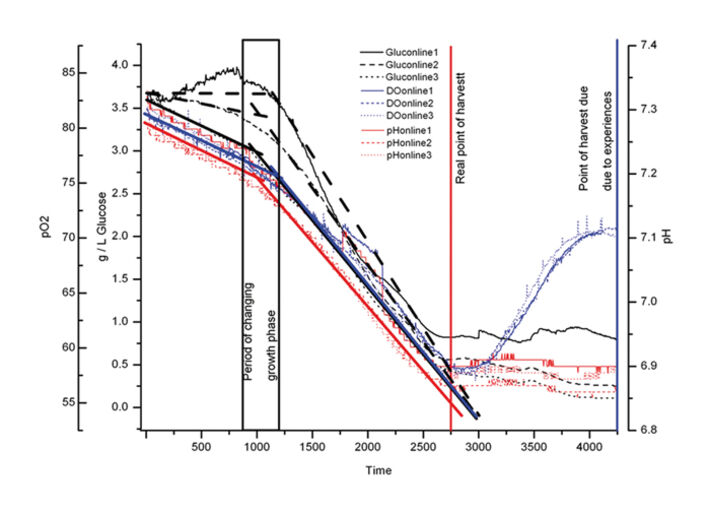
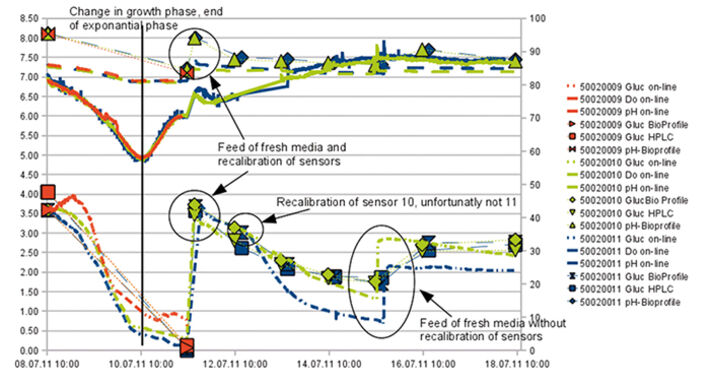
The starting glucose concentration measured by HPLC (4.05 g L-1) and enzymatic analyzer (3.59 g L-1) showed a difference of 12.5 %. As the glucose sensors were calibrated according to the values of the enzymatic analyzer the inline glucose sensors showed initial values of 3.6 g L-1. One of the glucose sensors showed slightly higher values after 4 days, but all the sensors detected the same glucose consumption (Fig. 2). The difference in initial glucose values of 0.5 g measured with the offline methods shows how difficult it is to conduct reliable glucose measurements. Depending on the method used, sampling, and time differences until analysis the values can get falsified. For this reason offline glucose measurement might not be suitable in process control. The same could be observed for pH measurement. Offline measurement showed an initial pH value of 8.1 while the online values measured with the chemical optical sensors at the bottom of each flasks were 7.2 - 7.3. This difference results from different environmental conditions during measurement. The incubator atmosphere is set to a CO2 concentration of 5 % v/v. CO2 needs to equilibrate in the media and the final pH is only reached after having the cell culture media in the incubator for a couple of minutes. This is why offline values taken at laboratory environment differ from online values. Online measurement always reflects the real situation. In the first four days the three typical growth phases showed in the response of all three sensors (glucose, pH, and pO2) (Fig. 3). For the first 10 hours there was clear lag-phase, in which the slope of all three sensors was different from the slope during the exponential growth phase that followed. In the next 36 - 42 hours the online oxygen sensor displayed the increased oxygen uptake during exponential growth, while during production phase the oxygen level in the culture media increased again, as oxygen transfer into the media exceeded oxygen consumption by the cells (Fig. 4). From experience and external sample analysis it was assumed that the cell culture needs at least 72 hours to reach the maximal cell density with a high level of living cells. In this test the online sensors detected the optimal point of harvest after approx. 45 hours. The used media slightly changed which resulted in faster cell growth. By conducting a time-controlled process or performing manual sampling these changes might never have been discovered, and a lot of production time wasted.
Conclusion
The application of inline glucose sensors in combination with chemical optical pH and pO2 sensors allowed continuous monitoring of these important process parameters in shake flask culture. Results obtained in these tests show the effectiveness of the online monitoring devices to control process quality. Compared to offline methods these sensors offer many advantages as no manual sampling is necessary, the risk of contamination reduced, and there are no reagent costs as with batch analyzers. Changes in the process can be detected immediately. The online DO reading and glucose measurement precisely show metabolic changes in the culture. With the integrated sensors measurements under real conditions can be obtained. The monitoring devices tested here were applied in shake flasks but might as well be applied for larger volumes like single-use bioreactors (S.U.B.).



