Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Assessing the Impact of pH Fluxes between the Sediment-Water Interface on the Overlaying Water Column off Namibia
Spatial variability of pore water pH investigated with single-use flow-through cells pH
Anita Flohr, Tim Rixen, Matthias Birkicht
Leibniz Center for Tropical Marine Ecology, Bremen, Germany
Eastern boundary upwelling systems, such as the Benguela upwelling system off the south west African coast, are characterized by high concentrations of dissolved inorganic carbon (DIC) and low pH in coastal surface waters. Only little is known about the role of DIC fluxes between the sediment-water interface on the Namibian shelf which is characterized by high organic carbon (TOC) content of the sediments. We investigated the spatial variability of pore water pH applying prototype flow-through cells (FTC-SU-HP8) to study the impact of sedimentary organic carbon content on the pH in pore water and the overlaying bottom water. The preliminary results suggest that the bottom water gets enriched with DIC while passing the shelf. A concomitant increase of total alkalinity (TA), likely caused by a combination of carbonate dissolution and e. g. denitrification in the sediments and bottom water, elevates the capacity to buffer the pH decrease despite increasing DIC concentrations.
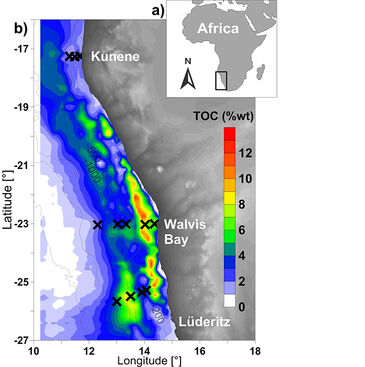

Coastal upwelling systems, such as the Benguela upwelling system (BUS) off south-west Africa, are regions where deep CO2-enriched water is brought to the surface reflected in enhanced CO2 emissions to the atmosphere, low pH concentrations and low carbonate saturations [e. g. 1]. Before reaching the surface, the subsurface water crosses the shelf characterized by high total organic carbon (TOC) accumulation within the sediments (Fig. 1) [2]. Here, pronounced oxygen depletion and DIC generation due to aerobic and anaerobic remineralisation of the organic carbon has been observed [3, 4] suggesting the bottom water gets enriched with DIC compared to its initial signature. To study the role of DIC fluxes between the sediment water interface in further enriching the overlaying water column we sampled inorganic carbonate system parameters of the pore water (DIC, pH) along cross-shelf transects during a cruise on the RV METEOR in 2013/14 (Fig. 1). Changes in pore water pH were investigated applying prototype single-use flow-through cells (FTC-SU-HP8) suitable for measurements in low volume samples.
Materials & Methods
Sediment cores were taken using a Multi corer (MUC) equipped with PVC cores. The pore water was sampled immediately after retrieval of the cores using Rhizons (Rhizosphere Research Products B. V., Netherlands; 19.21.23) which were inserted into the cores through predrilled holes at 1 cm intervals (Fig. 2). The Rhizons were connected to the FTC (FTC-SU-HP8, PreSens) and a syringe (10 mL) by Luer-Lock connectors for collection of the pore water. The FTCs with integrated pH sensors were stored in seawater before and after usage. After retrieval of pore water, the Rhizons were de-connected from the FTC-syringe combination. Air was removed from the syringe, the FTC was rinsed and locked by a stopper. The FTC was connected to a fiber optic pH meter (pH-1 mini, PreSens), and pH was recorded until the values had stabilized. Afterwards the pore water samples were processed and preserved for later analysis of DIC and nutrient concentrations. Presented data comprise preliminary results of pH and total alkalinity (TA) (VINDTA 3S, Marianda, Kiel). To calibrate the VINDTA 3S and to account for accuracy and drift of the pH sensors certified reference material (CRM, batch #111, provided by A. Dickson, Scripps Institution of Oceanography, La Jolla, US) was used. On the basis of pH and TA the DIC was calculated (DICcalc) using CO2.sys software [5].
pH Measurement in Pore Water
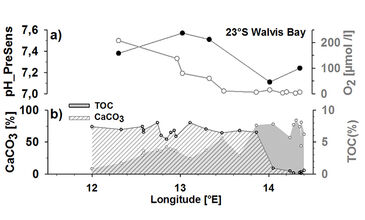
The variability of pH measured in the supernatant water of the cores correlates with O2 concentrations. Accordingly, on a spatial scale, lowest pH in supernatant and pore water of 1 cm below the surface were measured in cores obtained from TOC-rich sites where hypoxic to anoxic conditions prevailed (O2 < 5 µmol L-1, pH approx. 7.1) (Fig. 3) indicating DIC generation in the course of (an)aerobic decomposition of organic matter (OM). Nevertheless, the drop of pH measured in supernatant water is smoother than expected under increasing DIC concentrations due to a concomitant increase in TA. The correlation of salinity normalised TA (TA35) and DIC35calc observed in supernatant water varies between 2 - 1 indicative of CaCO3 dissolution (TA : DIC = 2) but also anaerobic remineralisation, e. g. denitrification (TA : DIC approx. 1) [6] known to occur in the oxygen minimum zone on the Namibian shelf [e. g. 7]. The generation of TA35 elevates the capacity to buffer pH reflected in a smoother decrease of pH at increasing DIC concentrations. It is in contrast to the oxygenated water column where the DIC35 increase during aerobic decomposition of OM is associated with a decrease in TA35 (TA : DIC approx. - 0.15) [8] and results in a steeper decrease of pH at increasing DIC concentrations. Denitrification relies on extremely low O2 concentrations which is a perennial feature of the bottom water on the northern BUS shelf [9] implying that the relative contri- bution of denitrification to the TA production in pore water and in supernatant water increases towards the coast.
Conclusion
The preliminary results emphasise the role of sedimentary fluxes in not only generating DIC but also TA which elevates the capacity to mitigate the drop of pH. To better understand the underlying processes optical pH sensors are more suitable to diplay and to account for the pronounced vertical variability observed in pore water pH, and the single-use flow-through cells pH proved most suitable for this application.
References:
[1] Feely, et al. (2008), Science, 320, 10.1126/science.1155676
[2] Inthorn, et al. (2006), Geology, 34(3), 205 - 208
[3] van der Plas, et al. (2007), African Journal of Marine Science, 29, 37 - 47
[4] Flohr, et al. (2014), Biogeosciences, 11, 885 - 897
[5] Pierrot, et al. (2006), ORNL/CDIAC-105a. Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, U. S. Department of Energy, Oak Ridge, Tennessee, 10.3334/DCIAC/otg.CO2SYS_XLS_CDIAC105a
[6] Chen, et al. (2002), Deep-Sea Research II, 49, 5365 - 5375
[7] Nagel, et al. (2013), Biogeosciences, 118, 361 - 371
[8] Broecker, and Tang (1982): Tracers in the sea
[9] Monteiro, et al. (2008), Deep-Sea Research I, 55, 435 - 450


