Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Development of Biomimetic Lung-on-a-Chip Platform
Oxygen transfer measurements with SensorPlugs at the inlet and outlet
Daniel Cheng, Charles Klassen, Harald C. Ott
IVIVA Medical, Beverly, MA, USA
The goal of this project was to provide a biomimetic lung-on-a-chip platform for tissue engineering and specifically for investigations of disease pathology. We first established long-term culture in a 3D lung model and measured baseline values for oxygen transfer and fluid filtration across the porous membrane in our model, both of which are known to be affected by viral infection in vivo. To measure oxygen transfer, we used PreSens' optical oxygen sensors placed directly in the fluid perfusion pathway both at the inlet and outlet of our scaffold. Using this setup, we have shown that our lung-on-a-chip model achieves similar rates of oxygen transfer, when normalized by gas exchange surface area, as that seen in commercial oxygenators.
The current COVID-19 pandemic highlights the critical need for a universally available, simple, affordable, scalable and physiologically relevant platform to study the effects of viral infection on the human lung. There is mounting evidence that an aberrant and maladaptive immune response, rather than the viral replication, contributes to acute lung injury seen in patients with severe disease. Given the heterogeneity in clinical outcomes it is possible that some patients mount a protective immune response, others injurious; therefore, any potential therapy to mitigate the role of immune system-induced injury needs to be individualized. To understand and modulate the immune response to SARS-CoV-2, a rapid screening test platform capable of reliably modeling the interaction between the virus and the immune response and its effect on the lung architecture and function is needed.
To date, there are no models that can accurately recapitulate the functional unit of distal lung tissue. Already existing lung-on-a-chip models contain microfluidic channel networks embedded in a non-biologic material, such as polydimethylsiloxane (PDMS), may not accurately recapitulate cell-extracellular matrix interactions, physiologic flow profiles, interstitial fluid shifts, and oxygen transfer seen in native tissue. Furthermore, non-biologic materials are prone to thrombosis and fouling, and thus may not be effective in studying pathogens that have extended incubation times requiring multiple days or weeks of culture.
At IVIVA, we have leveraged multiple technologies including 3D printing and thin film fabrication to develop a fully biologic scaffold with embedded vascular and airway networks separated by a thin (approx. 5 µm) porous gelatin membrane. Cells can be seeded on opposite sides of the membrane and maintained in an air-liquid-interface culture. In preliminary experiments, we have confirmed engraftment of various cells in our 3D scaffolds and maintained culture for over 21 days. We hypothesize that a physiologically relevant biologic scaffold will enable the study of the direct effects of viral infections on lung tissue as well as the immunological sequelae and enable the evaluation of therapeutic interventions. One of the most important functional metrics for evaluating functional lung tissue is oxygen transfer from the airway into the vascular blood supply.
Our first objective was to develop our lung-on-a-chip model and to establish functional baseline metrics for oxygen transfer and fluid filtration in an acellular model. These measurements will next be repeated in our scaffolds that have been seeded with endothelial cells and alveolar epithelial cells in the vascular and airway compartments, respectively. By first establishing the functional baseline measurements for oxygen transfer in an acellular scaffold, we can then measure the effects of cells and compare to known physiological values. We will then introduce a well-characterizing perturbation into the system, bacterial lipopolysaccharide (LPS), and measure its effects on gas transfer to validate our model.
Materials & Methods
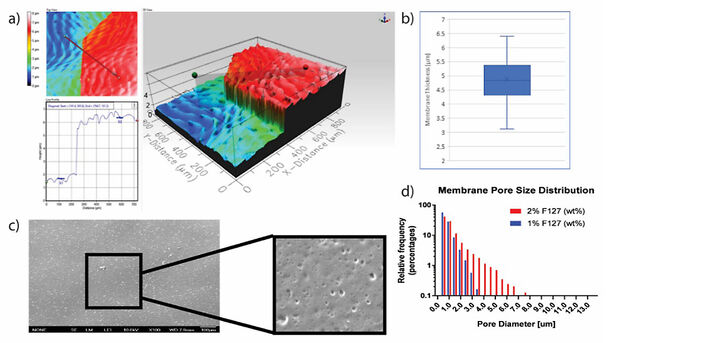
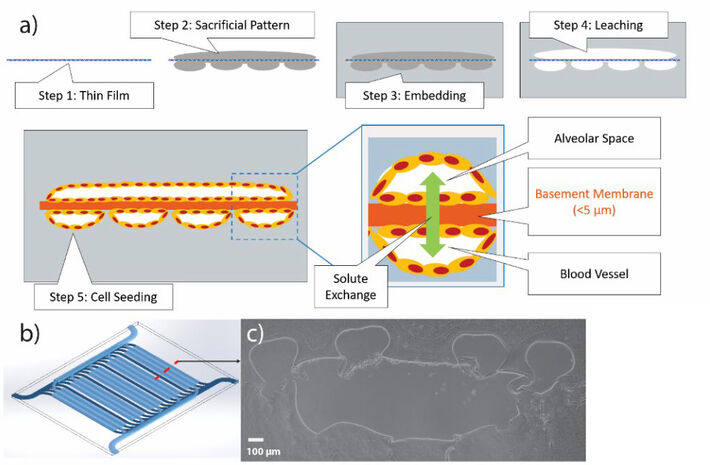
First we generated basement membranes of defined composition with controlled thickness of approx. 5 µm (Fig. 1a,b). By using F127 as a sacrificial porogen we created 0.1 - 4 µm pores, similar to those observed in the human bronchial basement membrane (Fig. 1c,d).
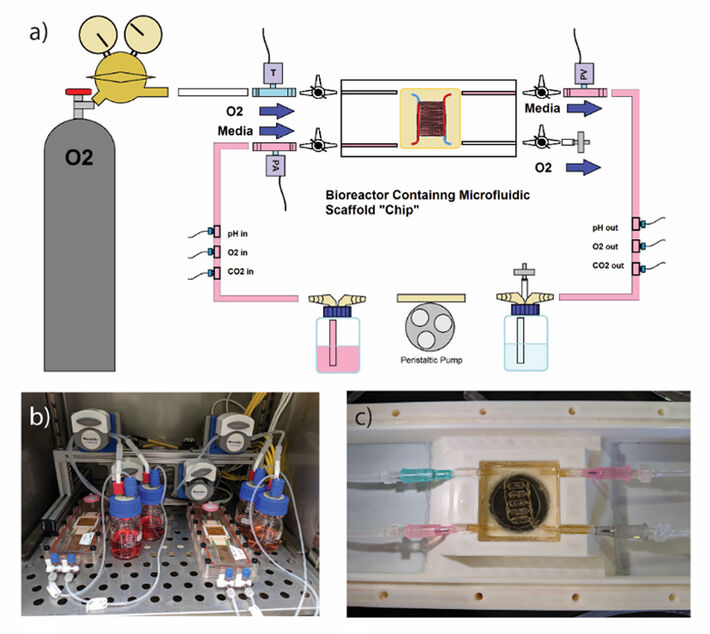
Then channels were 3D printed on both sides of the membrane again using sacrificial gel ink (Pluronic F127). Gelatin was cast onto these membranes using 3D-printed plastic molds and the gelatin was crosslinked using microbial transglutaminase before washing out the sacrificial channel patterns. After the F127 is evacuated, hollow channel networks remain that can be directly connected to flow, driven by a peristaltic pump, and perfused with fluid or air. The hollow channels can also be seeded with cells that will adhere to the gelatin channel walls and form a cell monolayer (Fig. 2a). Our channel geometry was designed with a 1000 µm wide alveolar space adjacent to smaller 200 µm vascular channels (Fig. 2b). The alveolar surface area is 232 mm2 and the vascular surface area is 184 mm2, a ratio of 1.26 (Fig. 2c). To establish an air-liquid interface (ALI) culture for this project, we perfused air through the airway channel network using a peristaltic pump with flow rates controlled using custom software developed in LabView. PreSens O2 SensorPlugs (together with pH and CO2 SensorPlugs) were placed in the fluidic pathway at both the inlet and outlet of the scaffold, with continuous fluid flow perfusing through the vascular network. There were also in-line pressure sensors for the trachea (T) airway inlet, the pulmonary artery (PA) inlet, and the pulmonary vein (PV) outlet (Fig. 3a). We perfused 100 % O2 through the airway network and measured the O2 transfer rate from the air channels across the membrane into the vascular network by subtracting the outlet O2 concentration minus the inlet concentration once the system had reached equilibrium at a given fluid and gas flow rate.
Results
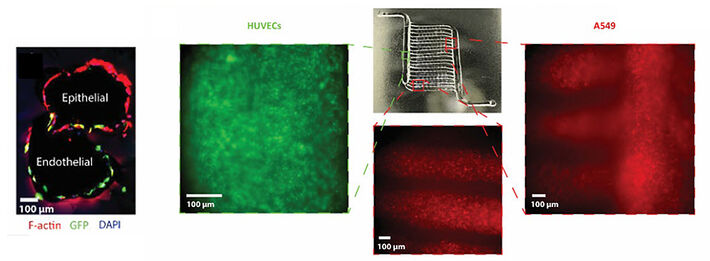
The lung's mechanical and biochemical environment greatly impacts cell function, yet existing in vitro models often do not accurately recapitulate physiology and often include synthetic materials. To address these challenges, we developed a fully biologic model based on a novel method to generate 3D channel networks lined by basement membranes. We have successfully cultured the generated scaffolds seeded with epithelial (A549) and endothelial (HUVEC) cells for up to 16 days in perfusion bioreactors with constant media perfusion culture (Fig. 4).
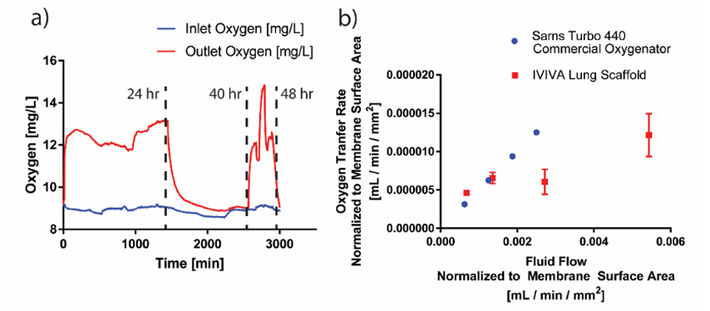
We have measured O2 diffusion and fluid transfer in our scaffolds and confirmed the structural integrity of the scaffold after perfusion with 100 % O2 and PBS at physiological pressures for 72 hours. Figure 5a shows measurement of oxygen concentration in a scaffold over the course of 48 hours using PreSens oxygen sensors at both the inlet and outlet of the vascular channel network. Total oxygen transfer through our scaffold is defined as fluid flow rate x (output O2 content - input O2 content). Media flow rate in the channels was varied, affecting O2 concentration over the time course. After 24 h of O2 perfusion, inlet O2 flow was decreased to 4 mmHg pressure overnight, then restarted the next day at 40 h. At 48 h, O2 flow was stopped.
After normalizing the O2 diffusion values to the surface area of the vascular network, we found that our O2 transfer rate was comparable to a commercial oxygenator, the Sarns Turbo 440 (Fig. 5b).
Conclusion
To date, we have developed a fully non-synthetic lung-on-a-chip model that can be used for air-liquid-interface cell culture. We have used PreSens O2 SensorPlugs to measure oxygen transfer form a gas-perfused airway channel network into a fluid perfused vascular network across a thin biologic membrane and found our scaffold to have similar oxygen transfer capabilities as a commercial oxygenator, when normalized by surface area. Future studies will be conducted to measure the effect of both vascular endothelial and alveolar epithelial cells lining the vascular and airway network channels, respectively, on oxygen transfer. We will then validate our system using the well-established perturbation of bacterial LPS, which is known to have a detrimental effect on cell survival and functional outcomes such as gas exchange and fluid transfer. Following validation of our platform, we are entering a collaboration with Massachusetts General Hospital (MGH) and a lab at the University of California San Francisco (UCSF) to study the effects of the SARS-CoV-2 virus on distal lung tissue. We will accomplish this by introducing a pseudotyped virus, a virus-like particle that is coated with the 'spike' protein from the coronavirus, into our lung model and measure its effects on functional outcomes such as oxygen transfer. This in vitro testing platform will enable direct observation to better understand the effects of the virus on lung tissue as well as the testing of potential therapeutic interventions on human cells prior to clinical studies.
Acknowledgment
This work was supported by grants from United Therapeutics and the Mendez National Institute of Transplantation Foundation.