Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Evaluation of an Optical CO2 Probe for Long-Term Monitoring in Stirred-Tank Bioreactors
Prototype Autoclavable CO2 Sensor Used in Low and High Cell Density Cultures
Melissa Hill1, Marcelo Kern1, Sayantan Bose1, Athanas Apostolidis2
1GlaxoSmithKline, King of Prussia, PA, USA
2PreSens Precision Sensing GmbH, Regensburg, Germany
When culturing mammalian cells, carbon dioxide gas is an important factor to stabilize the pH in the medium and to adjust the atmosphere to physiological levels for the respective cell line. CO2 levels in bioreactors fluctuate throughout the run and can affect cell growth, viability, and quality attributes if the levels get too high or low. CO2 levels are primarily monitored with offline measurements, so a need exists for a robust, accurate CO2 probe for constant monitoring.
The prototype optical CO2 probe by PreSens has a stainless-steel housing and a standardized thread so it fits standard bioreactor ports or port adapters. The sensor was integrated like other probes in a port at the bioreactor lid. As optical sensors are independent from the flow velocity of the sample, they are ideally suited for measurements in moving liquids.
We tested a novel optical CO2 sensor probe for cell culture monitoring in a stirred-tank bioreactor. The online measurements were compared to offline data obtained from Rapid Point and Nova Flex. A pilot run for 6 days at low cell density showed no drift in the CO2 signal and fluctuation in the measurement aligned with CO2 sparges. In a 14-day trial at high cell density, the probe showed larger deviation from reference measurements after day 8, which might have been caused by the high cell density. In the following 14-day trial at low cell density, the CO2 probe was recalibrated after day 5 as it trended a little lower than the offline data. After that, the online data were in good accordance with the reference measurements.
Material & Methods
The optical CO2 probe with a single-use opticap connected to the CO2-1 SMA meter was integrated via a port in the bioreactor lid of a 3 L glass reactor and autoclaved in 1X PBS prior to inoculation with a CHO cell line. Online CO2 measurements were controlled by the PreSens Measurement Studio 2 software and taken at 30 second intervals, or faster measurement rates when required such as the beginning of the run or during single-point calibration. The PreSens probe was one-point calibrated to either a Siemens RAPIDPoint 500 Blood Gas System Analyzer (Model # RP500) or the Nova Biomedical BioProfile FLEX Analyzer reading at the beginning of the tests. The online data was compared to offline readings determined with Rapid Point and Nova Flex.
Results
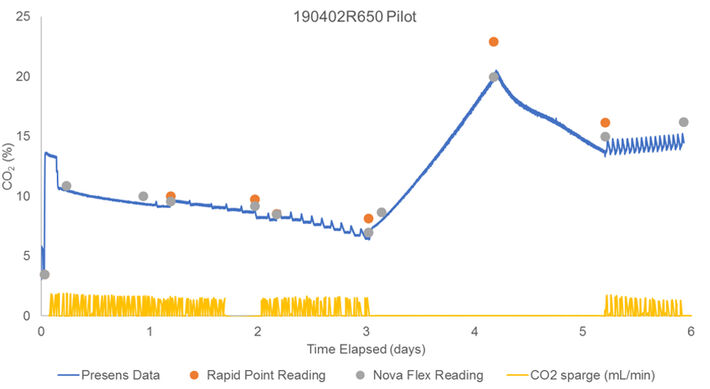
Figure 1 shows PreSens CO2 measurements and reference data for a pilot run in low cell density culture (6 million cells/mL) for 6 days. The pilot run was conducted to get a general overview of the optical sensor performance and possible drift. Prior to inoculation, the CO2 probe was single-point calibrated to the Nova Flex reading. The optical CO2 readings trended well with the Rapid Point data but were closer to the Nova Flex measurements. During the whole 6 days of the run, no drift could be detected. Fluctuations seen in the optical CO2 probes measurements aligned with CO2 sparges.
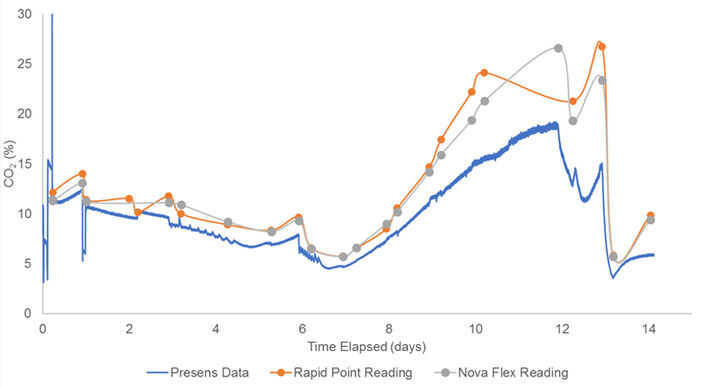
The following trial in high cell density culture (40 million cells/mL) lasted 14 days (Fig. 2). Calibration data from a previous run was used where the probe was single-point calibrated to the Rapid Point readings. Compared to reference measurements, the optical CO2 readings taken with the prototype probe trended a little low for the first 8 days, then the deviation increased for the remainder of the run. The high cell density most probably caused this deviation at this time. The high cell density likely changed the chemical environment of the reactor such that the initial calibration done on day 0 was not accurate. However, a single-point calibration on day 8 may have helped for the rest of the run.
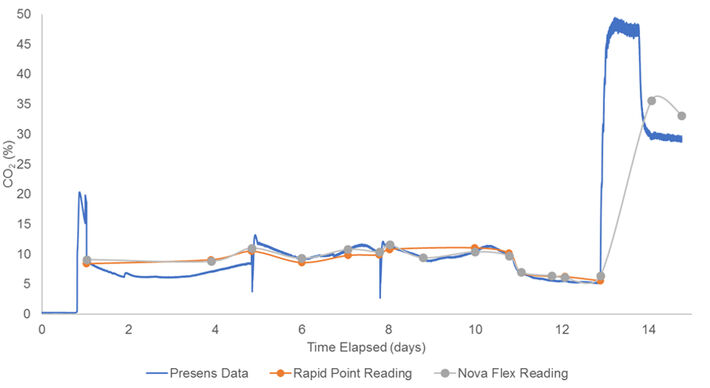
For the second 14-day run, a low cell density culture (6 million cell/mL maximum) was used (Fig. 3). The optical CO2 probe was again single-point calibrated to Rapid Point values at day 0. As observed for the previous trial, the optical measurements trended a little low until day 5. Another single-point calibration was conducted, which seemed to resolve the issue, and the optical measurements were in good accordance with the reference data for the remainder of the run. On days 4 and 7 the reactor was batched by removing some culture volume and replacing with fresh media to keep the cell culture low and healthy. Because of the temporary removal of cells and media, the recorded CO2 values temporarily dropped until the volume in the bioreactor was recovered. From day 13 until the end, the optical CO2 probe showed very high CO2 values. The CO2 levels in the culture increased rapidly as seen in the Nova Flex readings, so the maximum measurement range (> 25 %) of the optical sensor was exceeded.
Conclusion
Our experiments showed that the prototype CO2 probe was successfully used to monitor CO2 levels online in cell culture. After single-point calibration, its measurements were in good accordance (within 20 % difference) with offline determined reference values. However, finding the optimal time for a second single-point calibration during long-term measurements need additional optimization. While evaluation of the sensor for different cell culture applications would be desirable, our data suggest this technology was suitable for long-term monitoring of CO2 levels in stirred-tank bioreactors.
Acknowledgement
The project leading to this application has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under Grant Agreement n°777397. This Joint Undertaking receives the support from the European Union’s Horizon 2020 research and innovation program and EFPIA.
Disclaimer
The communication reflects the author's view and neither IMI nor the European Union, EFPIA, or any Associated Partners are responsible for any use that may be made of the information contained therein.




