Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
O2, pH and CO2 Dynamics in Salt Marsh Tidal Ponds
Investigations under Different Light Regimes with Optical Profiling Microsensors and an Automated Micromanipulator
Ketil Koop-Jakobsen1 and Martin Gutbrod2
1University of Bremen, MARUM - Center for Marine Environmental Scienes, Bremen, Germany
2PreSens Precision Sensing GmbH, Regensburg, Germany
Salt marsh ponds are extreme environments characterized by high microbial activity and strong biogeochemical gradients at the sediment water interface. In this study, we investigated the O2, pH and CO2 dynamics in the top sediment layer of marsh ponds under different light regimes. Using the Automated Microprofiling System from PreSens (Automated Micromanipulator AM & Profiling Microsensors PM) profiling of O2, pH and CO2 was conducted on marsh pond sediment cores in a growth chamber under light exposure ranging from 0 to 350 photons m-2 s-1. The oxygen penetration depth increased from 1 mm in darkness to 2 - 6 mm in light. At the sediment-water interface, photosynthetic activity by benthic microalgae and other photoautotrophs resulted in supersaturated oxygen conditions (400 - 700 μM) in light. In contrast, pH and CO2 at the sediment surface was unaffected by the varying light conditions and high photosynthetic activity suggesting a high buffering capacity of the pond water and sediment.
Salt marshes are important habitats making up the transition zone between the land and the sea. They provide a wide variety of ecosystem services such as coastal protection against storm surges and sea level rise, as well as nutrient retention and carbon sequestration. The surface of the salt marsh is inhabited by grasses, which are adapted to frequent tidal inundations and tolerant to variable salt concentrations. Shallow marsh ponds can be prominent features in the salt marsh landscape (Fig. 1). The ponds are generated by erosion typically after vegetation dieback or ice erosion, leaving a permanent pool of water on the marsh surface. The size varies, but usually the pools are 5 - 10 m in diameter and 20 - 50 cm deep. Marsh ponds function as refuge, shelter and spawning area for a large variety of fish and shellfish. In response to sea-level rise and increased storm events, the area coverage of ponds in tidal marshes are expected to increase, which may alter the marshes capability to sequester carbon and retain nutrients. So far, salt marsh ponds are often overlooked in salt marsh ecosystem studies, but they have recently gained increased attention in response to their expansion in area coverage. In tidal marshes, the ponds are frequently flooded (in the low marsh at ever tide, in the high marsh once or twice a month). For most of the day, the water is stagnant causing biogeochemical gradients to build up in the sediment and water column. The bottom of the marsh ponds is dominated by unvegetated muddy sediment usually with a thick microbial mat on top consisting of a diverse consortium of microorganisms, including green layers of benthic microalgae, and in these studies, a pink layer consisting of a consortium of bacteria utilizing the sulphur cycle was also shown (Fig. 2).
The marsh ponds are an extreme environment, fully exposed to sunlight, causing light and temperature in the pond to vary significantly throughout the day. Also, salinity can increase beyond ocean strength due to evaporation. The photosynthetic activity of benthic microalgae and other photoautotrophs and respiration of the microbial mat is responsible for marked spatiotemporal changes in O2, pH and CO2 over the course of the day. However, the daily variation in these ponds still needs to be characterized. In this study, we investigated the spatial dynamics of O2, pH and CO2 at the sediment water interface using microoptode profiling equipment from PreSens. The scope of the study was to elucidate how benthic microbial activity affects small-scale vertical biogeochemical gradients under different light regimes.
Material & Methods
Using the microprofiling optode system from PreSens (Automated Micromanipulator AM and Profiling Microsensors PM), the spatial oxygen, pH and CO2 dynamics were investigated under different light regimes: 0, 25, 150 and 350 photons m-2s-1 photosynthetically active radiation (PAR). Sediment cores (∅ 7 cm) were collected in a marsh pond in the salt marsh of the Plum Island Estuary, Northern Massachusetts, USA. The microprofiling was conducted in a growth chamber at a constant temperature of 20 °C (Fig. 3). Vertical profiles were measured from 1 cm above to 1 cm below the sediment surface with a spatial resolution of 150 - 500 μm per step. Oxygen and pH profiles were measured in 3 replicate cores, whereas CO2 concentration profiles were measured in two selected cores. The position of the sediment surface was determined by visual inspection.The overlying water was stirred by letting air stream over the water surface.
Microprofiling Results
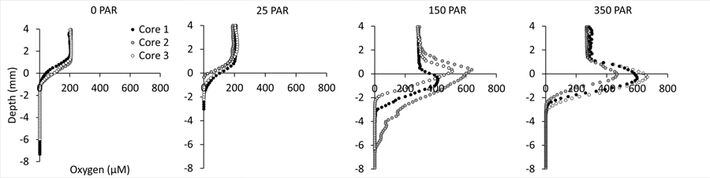
The Profiling O2 Microsensor facilitated detailed insight into the oxygen dynamics at the bottom of the tidal ponds, which proved to be highly variable with changing light conditions (Fig. 4). In darkness and at very low light conditions (25 PAR), the sediment became anoxic within the first millimeter. In light (150 and 350 PAR), however, the photosynthetic response of the microbial community was clearly shown in the oxygen profiles reaching an oxygen concentration at the sediment surface of 400 - 700 μM, which is 2 - 3 times higher than the atmospheric oxygen saturation (230 μM O2 at 20 °C and sal. 35 PSU). The peak in oxygen concentration at the sediment surface did not vary between 150 and 350 PAR indicating, that the photosynthetic activity of the microbial community became light saturated at very low light intensities. The photosynthetic activity had a marked impact on the oxygen penetration depth in the sediment, which increased from approximately 1 mm in darkness to 2 - 6 mm at 150 PAR.
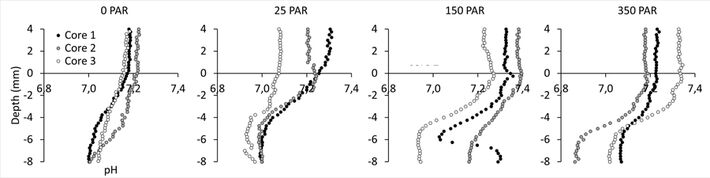
pH of the water column was relatively stable at 7.2 - 7.4. From the sediment surface to a depth of 6 - 8 mm pH slowly declined by 0.2 pH units. Although the oxygen profiles showed a marked photosynthetic activity during light exposure this activity did not affect pH at the sediment surface, and no spatial differences in the pH profiles were observed among different light treatments (Fig. 5).
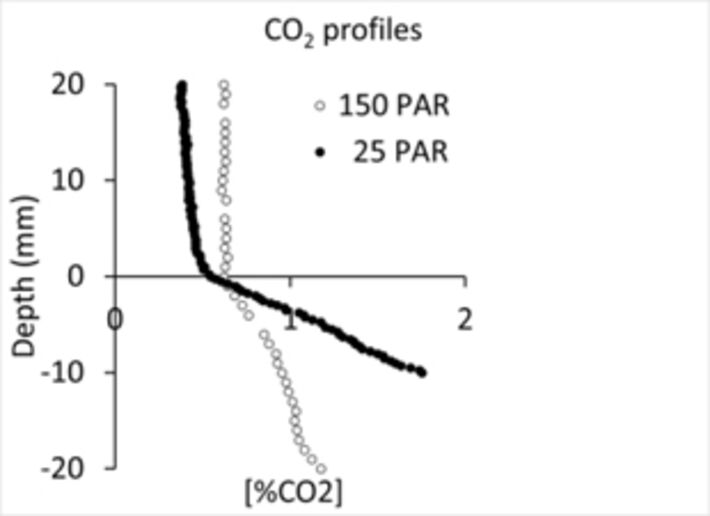
CO2 was measured in selected cores at light intensities of 25 and 150 PAR. The CO2 concentration was stable in the water column, but increased significantly through the sediment, in response to CO2 accumulation facilitated by benthic respiration and supported by lower pH conditions (Fig. 6). To our surprise the large benthic photosynthetic activity markedly affecting the O2 profiles had no effect on the pH or CO2 concentration at the sediment surface, which suggests that the marsh pond water has a high buffer capacity.
Conclusion
The microoptode profiling system enabled profiling at the sediment water interface with a high spatial resolution, giving insight into the biogeochemistry of this extreme environment. The system allowed an easy adaption to different core dimensions and interface heights. The fast response time of the O2 and pH microoptode accomodated fast profiling within a time-frame of 20 - 40 min per profile. The CO2 optode has a longer response time and profiling would usually take a couple of hours. Furthermore, CO2 measurements were complicated by interference from high sulphide concentrations in the pore water.