Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
SFR vario Discriminates Good from Bad in Shake Flasks
Online Biomass and O2 measurements help optimize screening conditions
H. Morschett
Microbial Bioprocess Lab - Helmholtz Innovation Lab, Institute of Bio- and Geosciences, IBG-1: Biotechnology, Foschungszentrum Jülich GmbH, Germany
Shake Flasks are widely used for screenings of production strains. We used the SFR vario for simultaneous online monitoring of biomass and oxygen in GFP producing C. glutamicum shake flask cultures. Tests at different working volumes and with different flask designs were carried out to analyze the impact on biomass development and product yield. In non-baffled shake flasks higher filling volumes caused faster oxygen depletion and resulted in less product yield. After a metabolic switch, these cultures entered a linear growth phase during oxygen limitation, which was recorded in biomass measurements. Measurements in baffled shake flasks showed better oxygen transfer into the medium and the cultures returned approximately the same product yield at all filling volumes. They reached maximum biomass faster, than in non-baffled flasks.
Microbial production processes, of e.g. recombinant proteins or organic compounds, are indispensable for the pharmaceutical and chemical industries these days. Screenings for ideal production strains are often carried out at shake flask scale, for easy parallelization and handling. In order to select a suitable production strain optimal growth conditions must be provided. However, monitoring technologies are not widely used at this scale so far.
In order to evaluate and efficiently optimize the screening conditions for C. glutamicum production strains we applied the SFR vario by PreSens. With this device pH, dissolved oxygen, and biomass can be measured simultaneously and continuously throughout the cultivation. The optical measurement principle allows recording the parameter values through the transparent bottom of the flask. We used 500 mL flasks with and without baffles and different filling volumes to investigate the different aeration properties and their effect on biomass development and product yield in C. glutamicum culture.
Materials & Methods
GFP producing C. glutamicum ATCC13032 [1] was cultivated in CGXII minimal medium with 7.5 g/L glucose, 7.5 g/L lactose, and 0.25 mM IPTG. All cultures were inoculated with an initial OD600 of 0.2. Cultivations were performed in 500 mL shake flasks with or without baffles at different filling volumes (25 mL, 50 mL, 75 mL and 100 mL). Cultures were placed in a Multitron Pro shaking incubator (Infors) at 30 °C, 25 mm shaking diameter and 210 rpm (shake flasks with baffles) or 150 rpm (shake flasks without baffles). Oxygen and biomass were monitored simultaneously and online with four SFR vario shake flask reader (PreSens GmbH).
Results
Figure 2 shows the oxygen and biomass measurements with SFR vario in non-baffled shake flasks. Cultures at all filling volumes reached oxygen limitation between 9 and 14 h. Higher filling volumes showed faster oxygen depletion and it took longer for oxygen levels to rise again. The biomass measurements all show a small peak followed by a short decline and then a linear increase in the biomass signal. The peak represents a morphological shift when oxygen is depleted, followed by a linear growth phase under oxygen limitation. The duration of the linear growth phase correlates with the flasks' filling volume. The thin medium layer in the flask with 25 mL volume could have caused the low biomass signal.
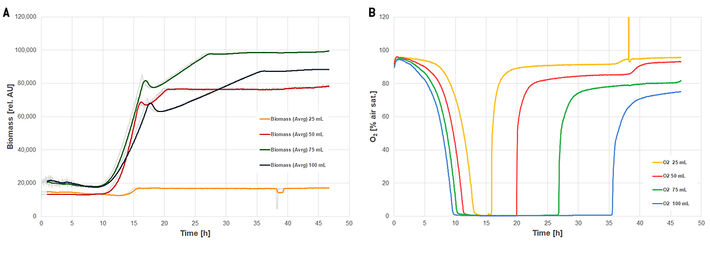
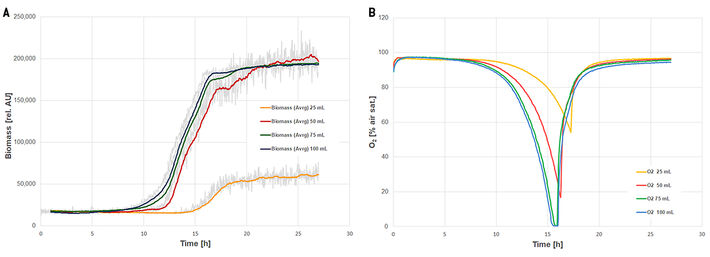
Figure 3 shows the SFR vario online measurements in baffled shake flasks. Like in the flasks without baffles, the thin medium layer at 25 mL filling volume seemed to cause low biomass readings. The cultures at 75 mL and 100 mL filling volume briefly reached oxygen limitation after 15 h. All cultures, except the 25 mL culture, show a characteristic kink in the growth curve at about half the cultivation time. (In the 25 mL culture this was not detectable because of the high noise in the biomass readings.) A metabolic switch from glucose consumption to consumption of lactose caused this kink. This switch cannot be seen in the oxygen readings, where a short rise in oxygen levels should have been detected during this phase. IPTG was added to the medium to induce GFP production, but it also induces the lac-operon. Therefore, lactose-hydrolyzing genes were active prematurely and lactose was split to galactose and glucose parallel to glucose consumption. Strictly speaking, the kink in the growth curve represents a metabolic switch from glucose consumption to the consumption of galactose.
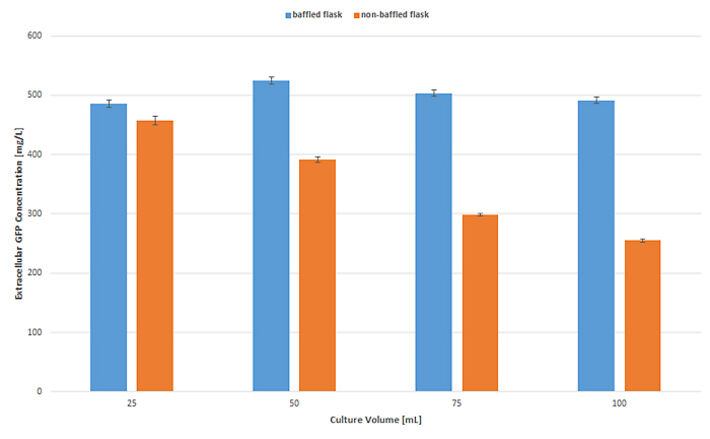
Analysis of GFP concentrations (Fig. 4) after the cultivation runs showed that in baffled shake flasks the different filling volumes have no significant influence on product yield. Cultures at all filling volumes produced about the same amount of GFP. In non-baffled shake flasks, on the other hand, GFP production was strongly affected by oxygen limitation. Even at 25 mL filling volume less GFP was produced compared to the culture in the baffled shake flask. With higher filling volume, the produced protein concentration significantly decreased.
Conclusion
We conducted these experiments with C. glutamicum cultures in shake flasks in order to optimize our screening conditions for production strains. Simultaneous oxygen and biomass monitoring with the SFR vario gave us detailed information about culture development, and the times when oxygen limitation or maximum biomass were reached. This allows us to adapt cultivation conditions and cultivation times accordingly, when comparing different strains in the future. The data obtained with the SFR vario is indispensable, if different strains need to be compared and helps building more reliable routines at shake flask scale.
References:
[1] L. Freier et al., ‘Framework for Kriging‐based iterative experimental analysis and design: Optimization of secretory protein production in Corynebacterium glutamicum’ Eng. Life Sci. (2016) 0, 1 - 12
[2] C. Keilhauer et al., ‘Isoleucine synthesis in Corynebacterium glutamicum: molecular analysis of the ilvB-ilvN-ilvC operon’ Journal of Bacteriology, Sept. 1993, 5595 - 5603



