Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Monitoring Oxygenation in Microfluidic Cell Culture Using 2D Sensor Foils as Growth Substrate (full text)
Oxygen Imaging with VisiSens™
C. Schmittlein1, R. J. Meier2, L. Sauer1, G. Liebsch2, J. Wegener1,3
1Institute of Analytical Chemistry, Chemo- and Biosensors, University of Regensburg, Regensburg, Germany
2PreSens Precision Sensing GmbH, Regensburg, Germany
3Fraunhofer Research Institution for Microsystems and Solid State Technologies (EMFT), Regensburg, Germany
White Paper Summary
Cells respond in very different manner regarding their microenvironment oxygenation. The oxygen availability for cells in microfluidics is strongly restricted due to the small volumes used. Here we demonstrate how the oxygen availability and the oxygen consumption during culture can be monitored label-free in microfluidics using the PreSens VisiSens™ oxygen imaging system.
Our study shows, that oxygen can be measured directly inside microfluidic channels via the use of luminescent sensors integrated into the growth substrate and as bottom plate for microfluidic channel structure. This label-free measurement technique enables readings in very small volumes and without consumption of the analyte during the measurement. Planar sensor foils enable a quantitative 2D read-out with the option to analyze local oxygen concentrations and gradients in any field of view, compare different regions or monitor several experiments in parallel channels at once.
In static conditions for a confluent MDCK II cell layer, the microfluidic chips were found to be depleted from oxygen within as short as 2.5 hours after fresh media was introduced. This fact was used to perform quick respiration tests, that allow for studying the effect of different drugs on cellular respiration in an intermittent flow experiment.
With respect to the oxygenation of microfluidic cell culture devices, the balance of oxygen influx and consumption by respiration needs to be tightly monitored and adjusted, since most materials used for making microfluidic structures have only a very limited permeability to oxygen. The data in this study shows how quickly the cell microenvironment turns hypoxic under static culture conditions with large impact on viability and proliferation of the cells under study. Control of O2 levels and well-adjusted perfusion is mandatory in those systems to get as close as possible to physoxic cell phenotypes and relevant read-outs of cell-based assays.
Ever since the advent of animal tissue and cell culture in 1912 the dishes and later flasks used to grow and maintain cells in vitro have not changed significantly in their principal geometry. Nevertheless, in vitro culture of animal and human cells became a success story with applications throughout the entire life sciences. The introduction of microfluidic devices has changed the situation and brought entirely new concepts and options to the field, like growing cells in perfusion, working with very low numbers of cells, exposing cells to well-defined and variable shear stress or direct coupling to downstream analysis systems capable of analyzing the incubation fluid [1]. Microfluidic devices have been established as versatile platforms to mimic various cell culture conditions or microenvironments in 2D and 3D tissue models. Lately, they have been widely applied in such innovative fields as the analysis and identification of circulating tumor cells [2], stem cell research and µTAS devices for cell analysis. Microfluidics is the basis for organ-on-a-chip or even body-on-a-chip approaches as they allow placing the corresponding tissues in series or in parallel to each other with a fluidic connection similar to the situation in the body. Due to their compact architecture microfluidic chips offer many advantages compared to standard well plate culture, e.g. low fluid or sample volumes, good process control as well as fast analysis and response times. It is considered as an enabling technology for massive parallelization of cell-based assays, which makes these devices gain in importance for drug discovery in general and high throughput analysis in particular.
The oxygen availability in the cellular microenvironment strongly affects the cellular phenotype and metabolism. For instance, regenerative stem and progenitor cells are known to stay in a proliferative state when kept in their oxygen niche [3], whereas elevated oxygen levels may become toxic and decreased oxygen levels may activate cell signaling pathways for differentiation or apoptosis. The balance of oxygen influx and oxygen consumption determines the state of the microenvironment. Cells in normal tissue have their specific microenvironments with respect to oxygen, which are referred to as physoxia. It varies from compartment to compartment of the body and ranges from around 13 % (v/v) O2 in the lung alveoli down to almost 0 % (v/v) O2 in bone. Despite of its unconditional importance the oxygenation of the microenvironment is a largely overlooked aspect of most in vitro systems and is only addressed by experts in the field. The small volume in microfluidic chips and the potentially low availability of dissolved oxygen increase the importance of precise oxygen monitoring. But even under commonly applied cell culture conditions in the standard dishes and incubators, there is a considerable mismatch between oxygenation in a cell culture incubator and the physiological situation. Most cell culture is still conducted with an oxygen content of around 19.9 % (v/v) O2 in the incubation atmosphere, which results from 95 % (v/v) ambient air (approx. 21 % (v/v) O2) and 5 % (v/v) CO2, which is commonly used to balance the medium pH. These conditions are referred to as normoxia, oxygen levels below this are denoted as hypoxia. The latter is misleading since oxygen levels in the human body (physoxia, see above) are lower than in regualr cell culture incubators and therefore more hypoxic than normoxic.
Oxygen in Microlfuidics
Regarding the balance of oxygen influx and consumption, the influx is the most critical parameter in microfluidic devices controlled by medium volume, sample size and perfusion rates. All three of them are under experimental control whereas oxygen consumption by the biological organisms under study is not [4]. A detailed understanding about the oxygen tension inside microfluidic devices is essential to bring the in vitro conditions closer to real physoxia in cell culture and provide the basis for more reproducible and standardized results with maximal physiological relevance.
The 'gold standard' for measuring oxygen is the Clark-type electrode with an amperometric read-out. However, its use in microfluidics is rather limited or, oftentimes, not applicable at all. A Clark electrode creates a direct contact between the sample and the external environment increasing the risk of introducing contaminations. More seriously, the electrochemical read-out consumes oxygen during the measurement, which becomes substantial when very small sample volumes are investigated. The use of optical oxygen sensors overcomes these drawbacks. They rely on the principle of dynamic quenching of luminescent probes. They are small in size, may be manufactured in various formats and the signals are recorded in form of visible light, so that data acquisition from the outside through the transparent walls of microfluidic chips is easily accessible. Thus, the read-out of luminescence signals is compatible with closed dishes without any mechanical connection so that interference with the sample is minimized. Luminescent sensor dyes can be applied as free molecules, incorporated in small sensor particles or embedded in a matrix material as planar sensors. All these formats have individual advantages in ease of use, applicability, biocompatibility and analytical performance. Whereas molecular sensors or sensor particles are easy to use within microfluidic devices by adding them to the perfusion fluid, they are prone to several pitfalls and drawbacks. Most importantly, molecular indicator dyes as such - or after integration in nanoparticles - leak cytotoxic singlet oxygen that is generated during the measurement in situ and may have adverse effects on the cell culture and spoil the experiment. Furthermore, dyes and nanosensors are potentially susceptible to severe photobleaching which limits their lifespan as functional sensors. The direct contact between probe and cell is often ignored but the reason for increasing concern as molecules and particles may accumulate in specific organelles or cellular structures which may affect cell physiology and the analytical read-out. As a consequence, the cells under study cannot be used for further experiments since they are contaminated with the oxygen probe. The use of planar sensors with immobilized indicators overcomes all of these drawbacks when the cells are grown directly on the surface of the sensor. Under these conditions the distance between cell membrane and sensor is in the order of 20 to 200 nm only. The dyes are immobilized in a biocompatible matrix material that hinders singlet oxygen from getting in contact with the sample. Even though luminescent probes are used for the measurement the cells remain free of any label and thus can be reused. Planar sensors are designed not to show any probe leakage making them biocompatible and the protected environment within the matrix provides significantly improved photostability.
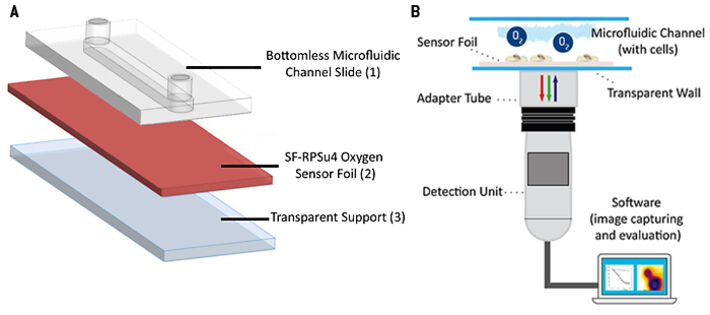
Planar optodes for oxygen can be used for inline monitoring when flow-through cells are connected to the inlet and outlet of a microfluidic device respectively. Alternatively, the sensors are deposited as spots within the channel or the reservoirs to monitor the oxygen content therein. For highest spatial resolution, planar O2 sensors are used as the bottom plate of microfluidic channel structures so that they serve as sensor and growth substrate for the cells under study at the same time. In combination with an appropriate camera, the set-up provides a 2D read-out over the entire fluidic structure. This 2D read-out enables visualizing gradients, as well as investigating various regions of interest in one experiment. In this study we present a 2D sensor read-out system, called VisiSens™, along with planar optical oxygen sensor foils integrated in microfluidic channels to investigate oxygenation in microfluidic cell culture. Due to (i) the small distance between membrane and sensor surface to be bridged by diffusion and (ii) the high O2-permeability of the cell membrane, the read-out mirrors the oxygen content of the cytoplasm in good approximation.
Materials & Methods
The oxygen sensitive planar sensor foil type SF-RPSu4 (PreSens, Germany) was immobilized upon a microscope slide using a silicone adhesive. The microscope slide provides a stiff, rigid and optically transparent support. A protection layer, present on the commercial sensor foil, was removed mechanically from the sensitive side of the foil to improve cell adhesion and to reduce the distance between cell and indicator dyes. A self-adhesive microfluidic structure without bottom plate (sticky-Slide I, 0.4luer; ibidi, Germany) was glued to the sensor foil providing the final microfluidic cell culture dish with integrated planar O2 sensor. The self-adhesive microchannel has a height of 450 µm, a channel volume of 113 µL and growth area of 2.5 cm2. In a further experiment, an 'open channel' system without channel top side closure was investigated for comparison. Therefore, an open PDMS-cultivation chamber with the same growth area of 2.5 cm2 and 1 mL of media volume was glued to the sensor foil. A lid was used to cover the chamber to prevent medium evaporation in the incubator. Read-out of the planar sensor was accomplished using the VisiSens A1 oxygen imaging system (PreSens, Germany). The read-out device is a USB-powered CMOS camera detector with incorporated excitation LEDs and optical filters. The sensor foil is excited using blue light and it returns red and green fluorescence emission. The luminescence of the red emitting probe is sensitive to the local oxygen concentration, whereas the green emitting probe is insensitive to oxygen and serves as internal reference to account for probe-independent impacts on emission intensity, like e.g. fluctuations in incident light intensity. The ratio of red and green signals provides an oxygen-dependent, ratiometric response in each pixel within the field of view. Two-point calibration of the sensor was accomplished applying cell culture media with 0 % (v/v) O2 (medium supplemented with 10 g/L Na2SO3 to generate an oxygen-free solution) and 100 % air saturation as the second calibration point. Automated time-series recording and evaluation of the oxygen images was done in the VisiSens AnalytiCal 1 software (PreSens, Germany). The complete set-up is sketched in Figure 2.
MDCKII cells were grown to confluence in the final microfluidic chip under static conditions according to the manufacturer's recommendations starting with a cell density of 1.0 x 105 cells cm-2 inside the microchannel. The regular growth medium was replaced by serum-free L-15 medium, the internal lab standard for assay buffers, at the beginning of the experiment. 2D oxygen recordings of cell microenvironment under static conditions in the chip were started 24 h after seeding, taking readings every 5 min. The medium was exchanged manually in regular intervals after the start of the oxygen measurement by slowly flushing 1 mL fresh L-15 medium through the channel.
Results
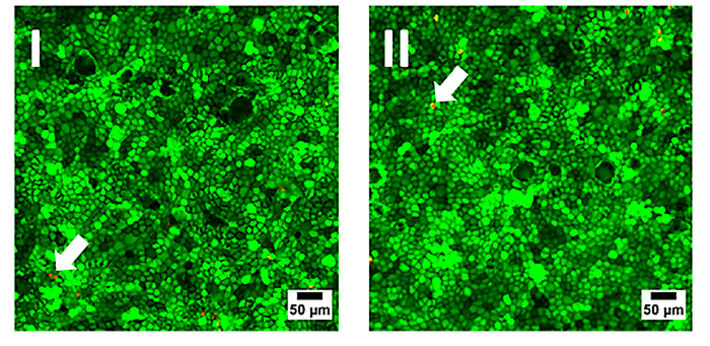
Initially, the biocompatibility of the sensor foils was investigated by seeding MDCK II cells in similar concentration into regular Petri dishes (reference) made from cell culture treated polystyrene and in Petri dishes with an oxygen sensor foil glued to the bottom of the dish. Cells were grown to confluence and cell viability on the different substrates was documented by an established live-dead staining (Fig. 3). This fluorescence-based assay contains two different probes: Calcein-AM and Ethidium-Homodimer. The former stains the cytoplasm of living cells brightly green, whereas the latter marks the nucleus of dead cells by red fluorescence emission due to intercalation of the dye into the DNA. Both samples show similar results with only very small amounts of red emitting dead cells in the monolayer (arrows in Fig. 3). The micrographs are interchangeable and there is no evidence for the presence of the sensor foil. Accordingly, the O2 sensor foils have proven their biocompatibility and can be used as a substitute for regular cell culture plastics.
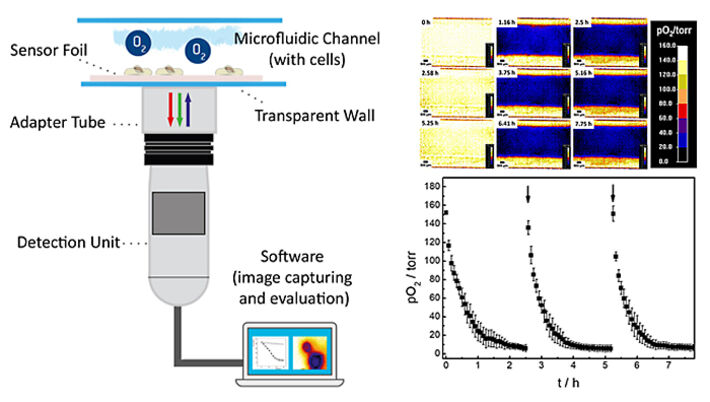
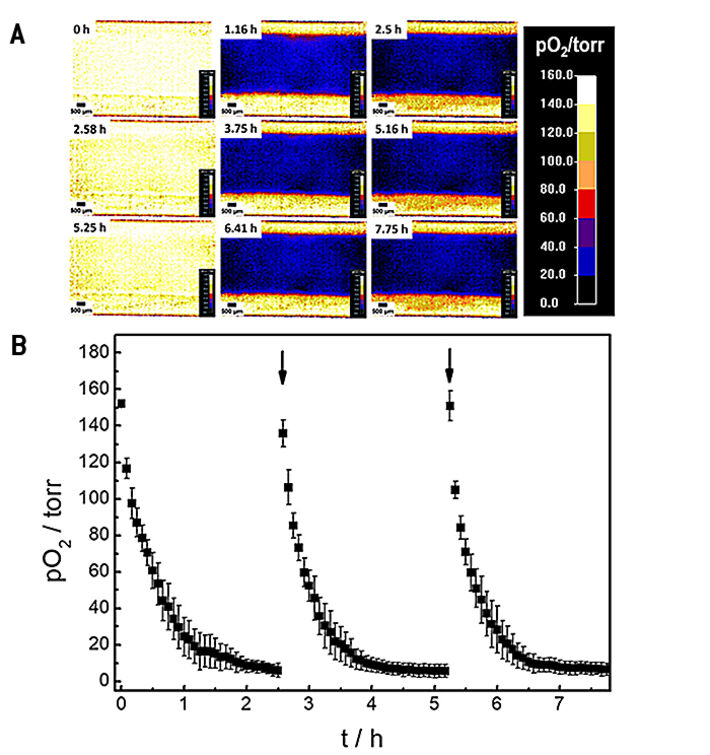
The oxygenation inside the microfluidic channel and thus, the microenvironment of the cells is investigated in a time series experiment with intermittent exchange of the assay buffer. Changes in 2D spatial oxygen distribution over time are reported as false color images in Figure 4A. The pseudocolors encode the respective oxygen levels in each pixel underneath the cell layer. The 2D 'oxygen images' were recorded by adjusting the adaptable field of view of the VisiSens A1 system to cover the whole channel width. An increased field of view leads to a decrease in image resolution and vice versa. The time series of images illustrates a laterally homogeneous, fast drop in oxygen levels within one hour as a result of cellular respiration in a non-perfused system made from materials (channel, bottom plate) with low oxygen permeability. The intermittent exchange of cell culture medium at 0 h, 2.58 h and 5.25 h leads to a repeated, complete and homogeneous channel reoxygenation.
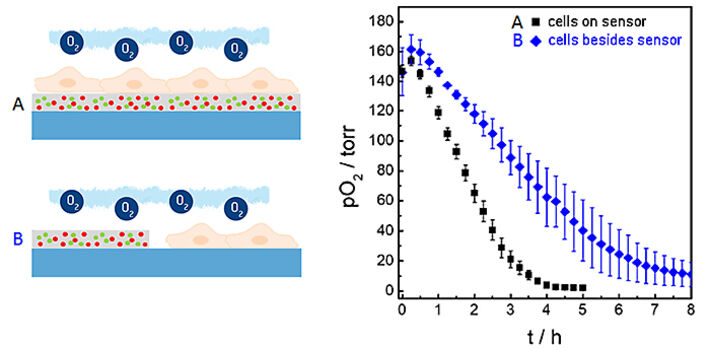
By selecting a region of interest (ROI) the time dependent changes in oxygen levels can be followed quantitatively as a function of time. Three consecutive oxygen consumption profiles of MDCK II cells cultivated inside the microchannel with intermittent flow, recorded in three individual measurements, are shown in Figure 4B. Upon addition of fresh L-15 medium, pO2 underneath the epithelial cell layer increases to normoxic values of around 150 torr. As cells in the channel consume oxygen without any significant O2 influx from outside the channel, a 50 % decrease of pO2 is observed surprisingly fast within (0.3 ± 0.1) h after exchange of L-15 medium. pO2 rapidly drops to hypoxic oxygen values below 10 torr within (1.5 ± 0.2) h after medium exchange. The time dependent oxygen profiles allow determining the oxygen consumption rates (OCR) of the cells under study for these exactly defined experimental conditions as will be shown in the next paragraph.
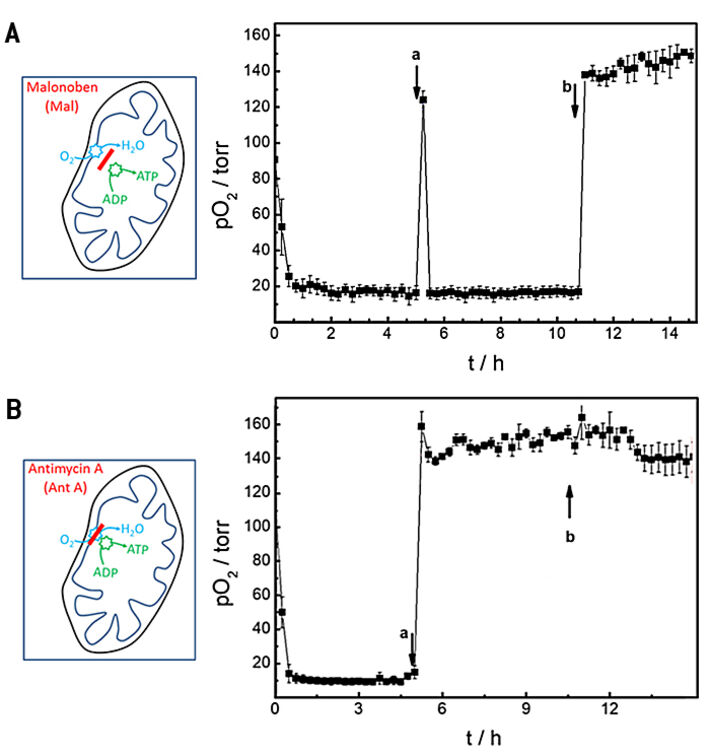
In further proof-of-concept experiments, we used an open channel system with integrated O2 sensor to study the effects of well-known drugs on the oxygen consumption rates of cells. Two drugs with established mechanisms of action for either uncoupling or blocking oxidative phosphorylation were studied with the composite device. Malonoben (Mal) acts as an uncoupler of respiration and ATP production whereas antimycin A (Ant A) blocks respiration. As a consequence, cells are known to increase their oxygen consumption rate when exposed to malonoben to compensate for the lack of ATP synthesis. In contrast, blocking of mitochondiral respiration by antimycin A ceases oxygen consumption entirely. The experiment was carried out in a consecutive three step procedure and the oxygenation in the chip was followed with time (Fig. 5). Initially, basal oxygen consumption was determined in standard L-15 medium, followed by a buffer exchange after 5 h to L-15 medium supplemented with 100 nM Mal (Fig. 5A) or 2 µM Ant A (Fig. 5B), respectively. Finally, cells were treated with 1 mL fresh L-15 medium containing 0.3 % (w/v) saponin, a detergent well-known to permeabilize cells. During baseline recording pO2 decreases due to cellular respiration to values below 20 torr within about 0.75 h after starting the measurement and OCR was determined to be (26.9 ± 0.2) pmol s-1 in L-15 medium (Fig. 5A). Replacement of L-15 medium by L-15 medium spiked with 100 nM malonoben (Fig. 5A a) is accompanied by transient reoxygenation of the channel to a value of (124 ± 5) torr. However, the uncoupling effect of Mal leads to an even faster consumption of the available oxygen by the cells in the channel as it is known from literature. pO2 decreased to values below 20 torr within 0.25 h. The fast oxygen consumption limits the precision of OCR determination which was calculated to (27.2 ± 0.1) pmol s-1 under these conditions. Addition of saponin to the incubation medium (Fig. 5A b) permeabilizes the cell membranes so that cellular respiration is irreversibly stopped. Accordingly, reoxygenation of the channel leads to time independent normoxy in the channel.
The basal oxygen consumption rate before the cells were exposed to the respiration inhibitor antimycin A (Fig. 5B) returned (28.3 ± 0.1) pmol s-1 in close agreement to values reported above. When oxidative phosphorylation is irreversibly blocked by Ant A, the reoxygenation of the channel due to medium exchange does not change anymore and remains at levels of (159 ± 9) torr. Addition of saponin does not have any significant additional impact as expected. Accordingly, OCR levels drop to values of (4.1 ± 5.2 10-2) pmol s-1. The big standard error indicates the limited precision of the measurement as hardly any oxygen is consumed. OCR in presence of saponin is formally determined to (0.3 ± 0.2) pmol s-1.
In the measurements described above we used the planar O2 sensor foils as sensor and growth substrate for the cells at the same time. Under these conditions the distance between the lower cell membrane and the sensor is in the order of 20 to 200 nm dependent on cell type. Due to (i) this small distance between membrane and sensor surface to be bridged by diffusion and (ii) the high O2-permeability of the cell membrane, this architecture provides a rather short response time of the sensor and the unique lateral resolution when it comes to imaging oxygen gradients. However, sensor foils can also be applied cell-free to probe the oxygenation of the microenvironment next to the cells. Figure 6 shows a comparison of the integrated sensor response when the cells under study are cultured directly upon the sensor surface as in the examples described above (black curve) or next to it (blue curve). The time-dependent decrease of pO2 shows significant differences in these two experimental situations. The reason for the slower decline of pO2 when the cells are grown next to the sensor foil is the extra diffusion distance between the respiring cells and the sensor as well as the differences in the liquid volumes that are being probed in either set-up.
Discussion and Conclusion
Our study shows, that oxygen can be measured directly inside microfluidic chips via the use of luminescent sensors integrated into the growth substrate. This label-free measurement technique enables readings in very small volumes and without consumption of the analyte during the measurement. Luminescent O2 sensors may be applied as small sensor spots for continuous one-spot-measurements or as planar sensor foils for laterally resolved studies, as shown here. Planar sensor foils enable a quantitative 2D read-out with the option to analyze local oxygen concentrations and gradients in any field of view, compare different regions or monitor several experiments in parallel channels at once.
With respect to the oxygenation of microfluidic cell culture devices, the balance of oxygen influx and consumption by respiration needs to be tightly monitored and adjusted, since most materials used for making microfluidic structures have only a very limited permeability to oxygen. The data in this study shows how quickly the cell microenvironment turns hypoxic under static culture conditions with large impact on viability and proliferation of the cells under study. Control of O2 levels and well-adjusted perfusion is mandatory in those systems to get as close as possible to physoxic cell phenotypes and relevant read-outs of cell-based assays.
References:
[1] Mehling M, Tay S (2014) Micorfluidic cell culture. Current Opinion in Biotechnology 25/95: 85-102
[2] Guansheng D, Fang Q, den Toonder JMJ (2016) Microfluidics for cell-based high throughput screening platforms—A review. Analytica chimica acta 903: 36-50
[3] Wahyu W (2014) Effect of oxygen tension on proliferation and characteristics of Wharton's jelly-derived mesenchymal stem cells. Biomarkers and Genomic Medicine 6(1): 43-48
Celeste SM, Keith B (2008) The role of oxygen availability in embryonic development and stem cell function." Nature Reviews Molecular Cell Biology 9(4): 285
[4] Brennan MD, Rexius-Hall ML, Elgass LJ, Eddington DT (2014) Oxygen control with microfluidics. Lab Chip. 14(22):4305-18