Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Cultivation of Bacteria and Mammalian Cells in Shake Flasks
Bacterial and mammalian cell suspension cultures are used in many ways e.g. in biotechnology for the production of compounds, and as model systems in fundamental research. Especially in medical research liquid cultures remain essential to facilitate the understanding and treatment of diseases. Learn about the pros and cons of suspension cultures, why it is important to monitor them tightly, what methods there are to measure it, and what possible setups PreSens offers.
Overview
For many applications cultivation takes place in liquid cultures. This method is used either for the enrichment of a distinct species or in order to obtain higher amounts of cell mass. Currently a vast variety of vessels for cultivating cells in suspension culture exist on the market e.g. Erlenmeyer flasks, spinner flasks, baffled flasks, roller bottles, and multi-well plates. For screening and process optimization in small scales aerobic microorganism and cells are commonly grown in shake flasks, because they represent a low cost and easy to handle opportunity to perform a large number of parallel experiments. In addition, shaking prevents sedimentation, biofilm formation and cell clumping and promotes an equal distribution of nutrients. The major drawback of conical flasks are the permanently changing cultivation conditions within the flasks, like nutrient availability, oxygen consumption, or accumulation of metabolic products. Overflow metabolism, pH changes and oxygen limitation often pass unnoticed in small scale flask cultures and lead to failures during follow-up scaling processes. Therefore, it is essential to monitor the state of the culture continuously.
Real-Time Measurements
PreSens offers the SFR vario and the SFR Shake Flask Reader, which are non-invasive measurement systems using optical-chemical sensor technology, that allow the determination of pH and oxygen in shake flasks. The SFR systems also record temperature and agitation speed (rpm), which could be convenient to future references during the analysis of data.
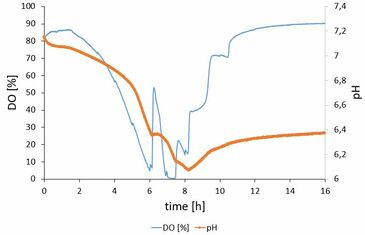
Figure 2 shows the time-course of a Escherichia coli K12 culture grown in an Erlenmeyer flask at 37°C using synthetic medium. Oxygen concentration and pH were monitored during the cultivation using the SFR vario.
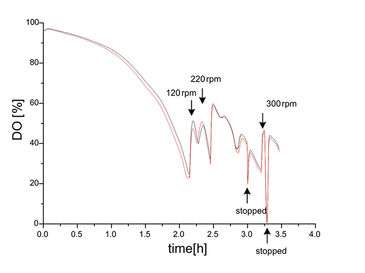
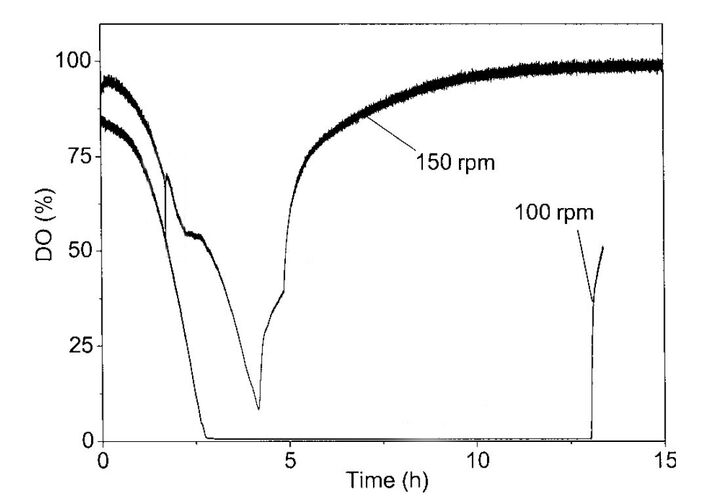
To counteract a noticed oxygen limitation you have to increase the surface of the medium, as oxygen entrainment only happens at the surface of the medium. Oxygen input relies on agitation of the surface of the medium to allow air to form small bubbles in the medium. Suitable measures include higher agitation speed, optimized ratio of medium volume to flask volume, and the use of baffled flasks. All these measures support oxygen input.
Figure 3 shows online oxygen measurements with optical sensors in two parallel E. coli DH5alpha cultures in LB complex medium. By constant monitoring and timely adjustment of the rpm oxygen limitations can be avoided. Online measurements enable a timely reaction and control the oxygen supply. At the same time it can immediately be verified that the taken measure had the desired effect.
Multi-Channel Set-Up for Shaking Cultures
To measure in Shake Flasks we offer two different systems (Figures below): The SFR vario which monitors and simultaneously measures O2, pH, biomass and optionally CO2, and the SFR Shake Flask Reader which measures pH and oxygen in up to 9 flasks. Up to 4 SFR varios can be controlled by the software and therefore, up to 4 flasks can be read-out in parallel. With the SFR Shake Flask Reader up to 7 systems can be linked together, resulting in a total of up to 63 flasks that can be monitored at once. Both systems offer automatic OUR calculation and monitor the analytes in Erlenmeyer flasks, cultivation tubes, or T-flasks.