Watch tutorials, webinars and informative videos about PreSens optical sensor systems.
Imaging of pH and pO2 on Irradiated Fibroblasts and Oral Squamous Carcinoma Cells
Dual read-out of pH and pO2 in 96 well plates with VisiSens TD
Yvonne Reinders1, Robert J. Meier2, Gregor Liebsch2 and Frank Haubner3
1Department of Plastic, Aesthetic, Hand & Reconstructive Surgery, University Hospital Regensburg, Regensburg, Germany
2PreSens Precision Sensing GmbH, Regensburg, Germany
3Department of Otorhinolaryngology, Ludwig-Maximilians-University Munich, Munich, Germany
One of the major challenges in radiation therapy is the interference with tissue repair processes. In this context, chronic wounds show hypoxic characteristics. However, sufficient oxygenation is mandatory for cell proliferation, granulation, migration, as well as protein synthesis during tissue repair. Additionally, pH is particularly important in signaling within damaged tissues and therefore precisely regulated. In this study, we investigated the effects of irradiation on different cell types in in vitro cell culture. We used biocompatible pH and O2 sensor foil equipped 96 well plates along with dual analyte 2D readout with the VisiSens TD system. Despite the usual use of 2D planar optode imaging systems with recording a contiguous planar sensor area, the technique is used in this study to read out a multitude of smaller sensor foils simultaneously in one recorded dataset. Furthermore, the system is able to detect signals from sensors with diverse sensor chemistry, in this case planar sensors for O2 and for pH, in the same experiment. Different sensitivities of fibroblast and oral squamous carcinoma cells were observed by complementing oxygen and pH differences with proliferation assays. This study highlights especially the distinct role of oxygen after irradiation and the difference in proliferation processes of irradiated normal dermal cells in contrast to irradiated tumor cells.
PreSens VisiSens TD Imaging Technique
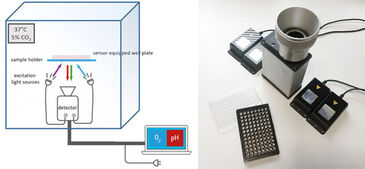
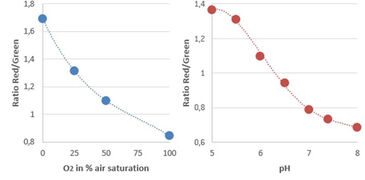
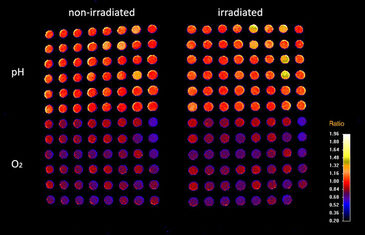
Experiments in this study were performed using the VisiSens TD system with the dual O2 and pH imaging software mode that is able to read out multiple O2 and pH sensor foils with one device simultaneously. Read-out of more than one well plate in one field of view was accomplished using the Big Area Imaging Kit accessory. The system was integrated in an incubator to perform the cell experiments under growth conditions (at 37 °C, 5 % CO2, non-condensing). For the cell measurements, the bottom of the wells of black 96 well plates were covered either with biocompatible O2 or pH sensor foils (SF-RPSu4 for O2 and SF-HP5R for pH). Each well plate consisted of 48 pH and 48 O2 sensor equipped wells. The sensor equipped well plates were β-irradiated prior to use. The well plates were placed on a Plexiglas plate that was mounted in the incubator, ensured optical transparency and held the well plates in position. The distance of detector lens and plates was adjusted to approximately 20 cm in order to simultaneously record two well plates in one field of view. The external light sources (Big Area Imaging Kit) were positioned crosswise and in dark field mode. The calibration solutions for the respective sensor foils were included in the same experiment in unused wells of the well plates. The calibration wells can be used to compensate potential signal drifts when e.g. measuring over long time periods, with temperature changes, or different radiation treatment of the sensors. For pO2 calibration triplicates of the calibration solutions Cal 0 (0 % O2) and Cal 100 (100 % air saturation = 19.85 % O2 at 5 % CO2, which was determined using air saturated cell culture media) were used. The calibration curve for pH was generated using 6 different 50 mM phosphate buffered media solutions from pH 5.0 to pH 8.0 and pipetting them in wells of each plate. Both used sensors materials matched the physiological relevant measurement range for cell culture.
Read-Out of pH and pO2
Read-out of the sensors was performed using the dual pO2 / pH read-out mode with use of the VisiSens ScientifiCal software. Images were taken every 30 minutes in an automated time series experiment over a time course of 140 hours. The software automatically takes images for O2 and pH at each time point. The recorded raw images of the pH and O2 time series are processed in the software resulting in ratio maps or calibrated analyte maps. A noise filter was used to exclude background signals. The mean values of the wells were read out by choosing regions of interest (ROI) over one well or several replicates. Time dependent data was obtained via the z-profiling or multi z-profiling tool in the software and exported to Excel.
Cell Culture and Irradiation of NHFs and OSCCs
Human dermal fibroblasts (NHF) were maintained in Dulbecco's modified Eagle's medium (DMEM; 4.5 g/L glucose) supplemented with 10 % FBS and 1% penicillin/streptomycin. The human oral squamous carcinoma cell line PCI4 was established from a male patient with a primary carcinoma at the root of the tongue. OSCC were maintained in DMEM (1.0 g/L glucose) supplemented with 10 % FBS and 1% penicillin/streptomycin. Cells were used for experiments at passages 3 - 6. The sensor foils in the wells were coated with FCS (1 hour at room temperature) for facilitating cell adhesion to the surface. 10.000 cells were used in each well. Experiments were conducted with n = 6. Irradiation of the cells was accomplished via an anterior portal by a 6 MV linear accelerator (3 Gy per min) at room temperature, as previously described by Pohl et al. [1]. Dosimetric evaluations were performed to guarantee a homogeneous dose distribution. The cells were irradiated with doses of 2, 12, and 20 Gy, respectively, as a low dose of 2 Gy is comparable to the standard dose typically applied to patients per irradiation and 12 Gy as a model for high dose. 20 Gy acted as a lethal dose and thus positive control. Non-irradiated cells under same conditions served as a control.
Effect of Irradiation on pO2 and pH of NHFs and OSCCs
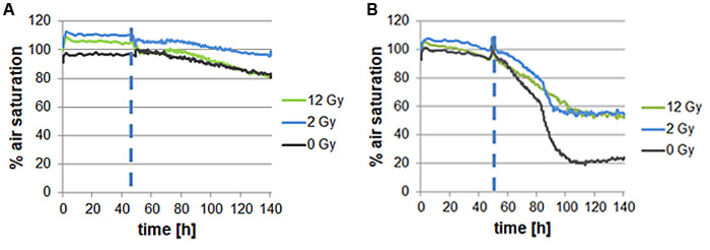
To gain deeper insight in the molecular processes of irradiation of cells, oxygen consumption of normal dermal fibroblasts as well as oral squamous carcinoma cells was analyzed. Thereby fibroblasts showed slightly decreasing oxygen consumption both when irradiated and not. This suggests only a minimal decrease in cell viability due to irradiation. In contrast, oral squamous carcinoma cells showed decreased oxygen consumption with increasing irradiation dose.
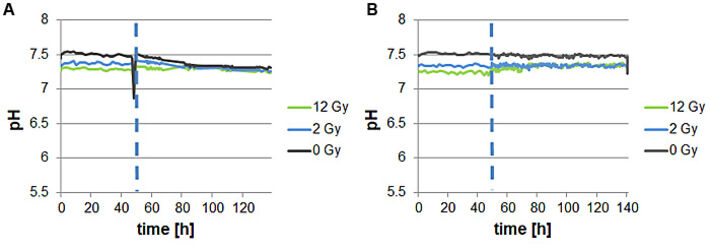
To complement the results of cells oxygen consumption measurements pH of normal dermal fibroblasts as well as oral squamous carcinoma cells was also determined. In contrast to the pO2 measurements, no differences of reaction on irradiation between fibroblasts and oral squamous carcinoma cells could be observed. All cell lines showed a rather stable pH value in time course after irradiation as well as without treatment.
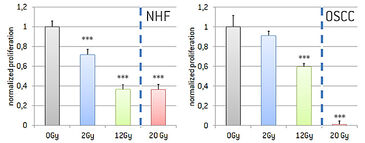
Effect of Irradiation on Proliferation of NHFs and OSCCs
To observe the effect of irradiation on dermal as well as cancer cells, fibroblasts and oral squamous carcinoma cells were grown in 96 well plates and irradiated with 2 Gy, 12 Gy, or 20 Gy, respectively. Proliferation rate was determined using a BrdU assay (Fig. 6). We were able to show that proliferation decreased with rising irradiation intensity. Highly significant effects for fibroblasts are depicted for 2 Gy, 12 Gy, as well as 20 Gy. Irradiation of oral squamous carcinoma cells had a reduced effect in comparison to fibroblasts. Nevertheless, also for oral squamous carcinoma cells a decreasing effect on proliferation which was significant for 12 Gy and 20 Gy was observed.
Conclusion
The VisiSens TD system is capable of detecting O2 and pH sensor foils in two 96 well plates. Growing cells directly on the sensor area allows for measuring the microenvironment of the cells rather than the media O2 and pH. This way, different irradiation treatments of various cell types in one conducted experiment and the repsective results can directly be compared. Furthermore, the calibration data can be recorded simultaneously during the experiment and therefore allows compensating for effects of the irradiation on the sensor response.
Reference
[1] Pohl, F.; Hassel, S.; Nohe, A.; Flentje, M.; Knaus, P.; Sebald, W.; Koelbl, O., Radiation-induced suppression of the Bmp2 signal transduction pathway in the pluripotent mesenchymal stem cell line C2C12: an in vitro model for prevention of heterotopic ossification by radiotherapy. Radiat Res 2003, 159, (3), 345-50